Current obesity treatments primarily rely on peptide-based GLP-1R agonists, which face challenges such as high production costs, the need for injections, and poor patient tolerability. The next generation of small-molecule GLP-1R agonists (such as Orforglipron) achieve oral bioavailability and are easier to manufacture at scale. However, these small molecules exhibit significant species differences—they selectively bind to the human receptor but not the rodent receptor—which has largely limited preclinical studies of their pharmacological mechanisms. A study published in Nature reveals the neural mechanism by which next-generation oral small-molecule GLP-1R agonists suppress feeding behavior through brain reward circuits. Using humanized mouse models, the researchers systematically investigated how small-molecule GLP-1R agonists regulate feeding behavior. The study focused on distinguishing the drugs’ differential effects on “homeostatic feeding” (eating driven by energy needs) versus “hedonic feeding” (eating driven by pleasure), and sought to clarify the specific neural circuit logic mediating these effects.
Model Construction: Humanization of a Key Receptor Residue
Researchers used CRISPR-Cas9 to introduce the S33W mutation at the mouse Glp1r locus, replacing serine at position 33 in the mouse receptor with the tryptophan found in the human receptor, thereby creating the Glp1rS33W humanized model. Experiments confirmed that this gene editing did not alter baseline metabolic rate, energy expenditure, or body weight, indicating normal metabolic function. In glucose tolerance tests (GTT), the traditional peptide liraglutide was effective in both genotypes, whereas the small-molecule agonists Danuglipron and Orforglipron showed glucose-lowering effects only in Glp1rS33W mice, demonstrating that the model successfully conferred sensitivity to human-specific small molecules.
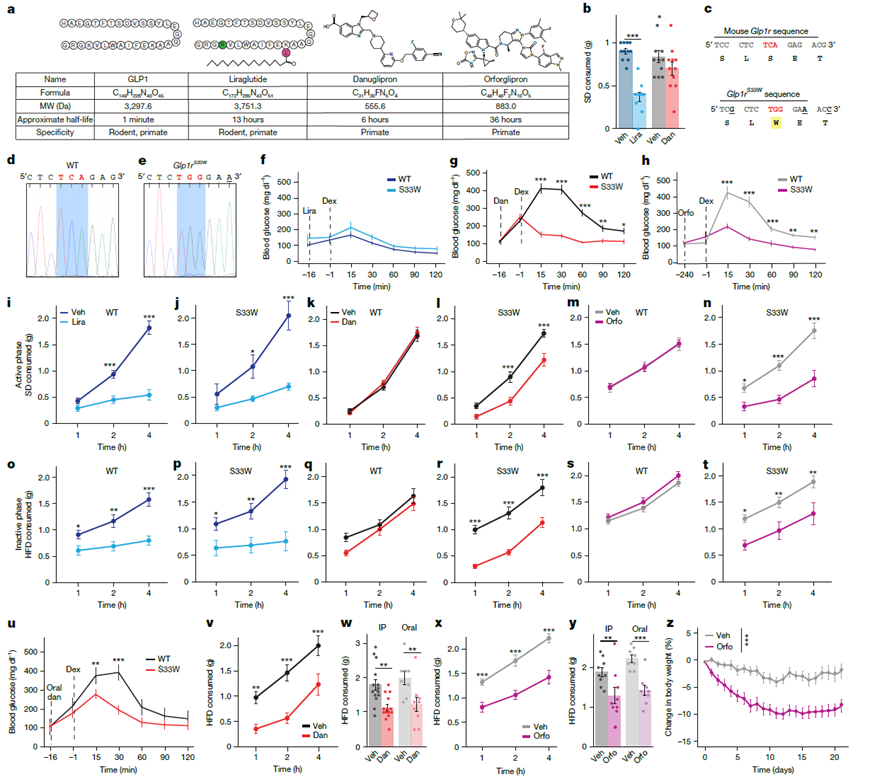
Figure 1. Validation of the small-molecule GLP-1R agonist-responsive mouse model
Distinguishing Feeding Suppression from Aversive Responses
Researchers employed machine learning-based pose estimation (SLEAP) and probabilistic classification models (Keypoint-MoSeq) for high-resolution behavioral dissection in mice. By comparing drug-treated groups with the emetic agent (LiCl) group and the satiated (Fed) group, they found that the behavioral profile induced by Orforglipron was markedly different from the “nausea-like” state induced by LiCl. Mice treated with Orforglipron reduced food intake while maintaining high levels of activity and exploratory behavior. In principal component analysis (PCA) space, their behavioral features clustered closer to the normal satiated group rather than showing behavioral suppression or aversion. This indicates that the drug’s weight-loss effects can be separated from pathways that produce discomfort.
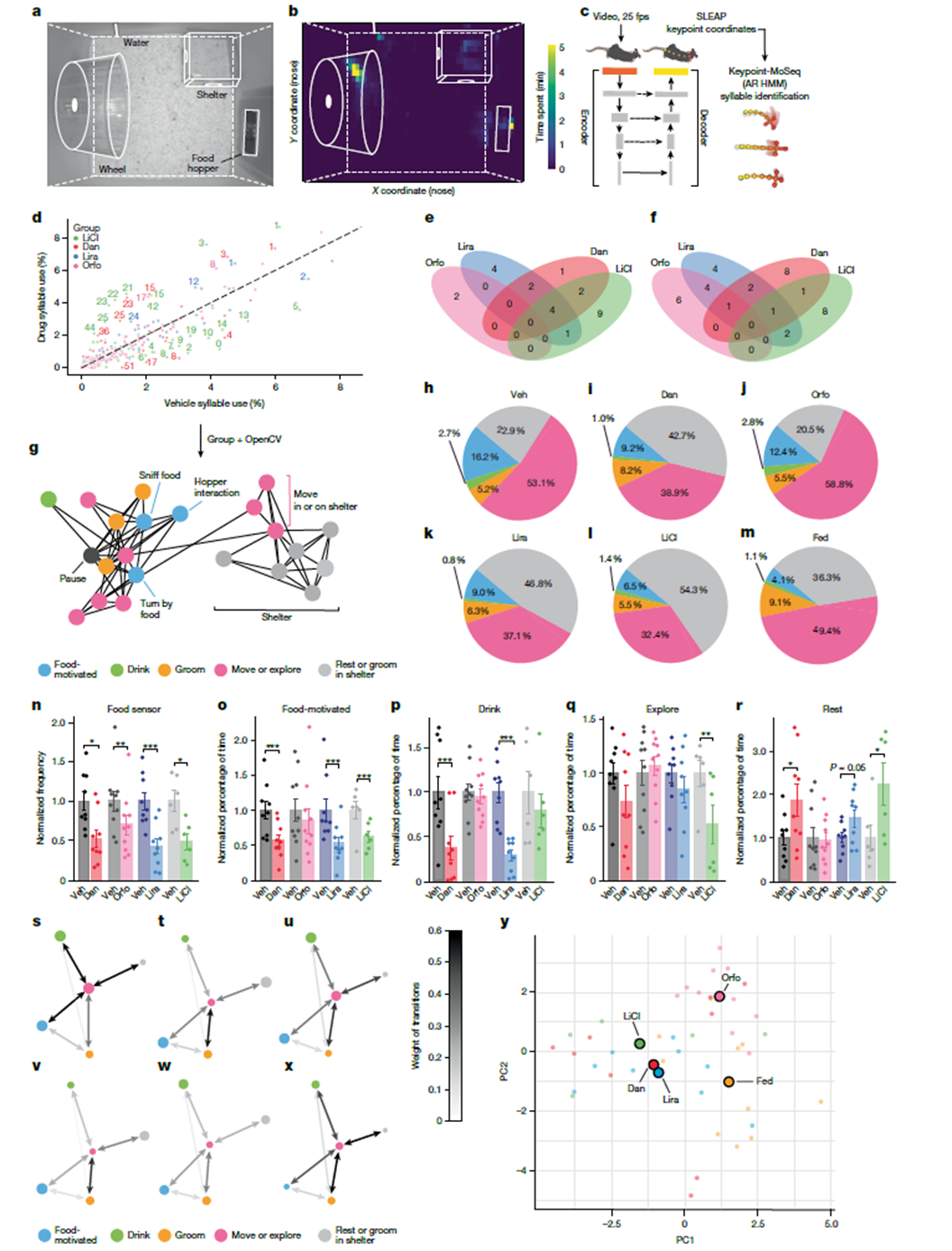
Figure 2. Machine learning-assisted behavioral analysis reveals distinct phenotypes associated with GLP-1 receptor agonists
Differential Activation by GLP-1R Agonists
By quantifying expression of the neuronal activation marker FOS, the study examined the drugs’ brain targets. Results showed that in Glp1rS33W mice, small-molecule agonists significantly activated the nucleus tractus solitarius (NTS), area postrema (AP), and central amygdala (CeA), but no obvious FOS induction was observed in the hypothalamic dorsomedial nucleus (DMH). Notably, Orforglipron produced a significantly higher ratio of NTS to AP activation compared with Danuglipron or liraglutide. This bias may explain differences in clinical tolerability among the drugs—a greater preference for activating NTS (satiety-related) over AP (aversion-related).
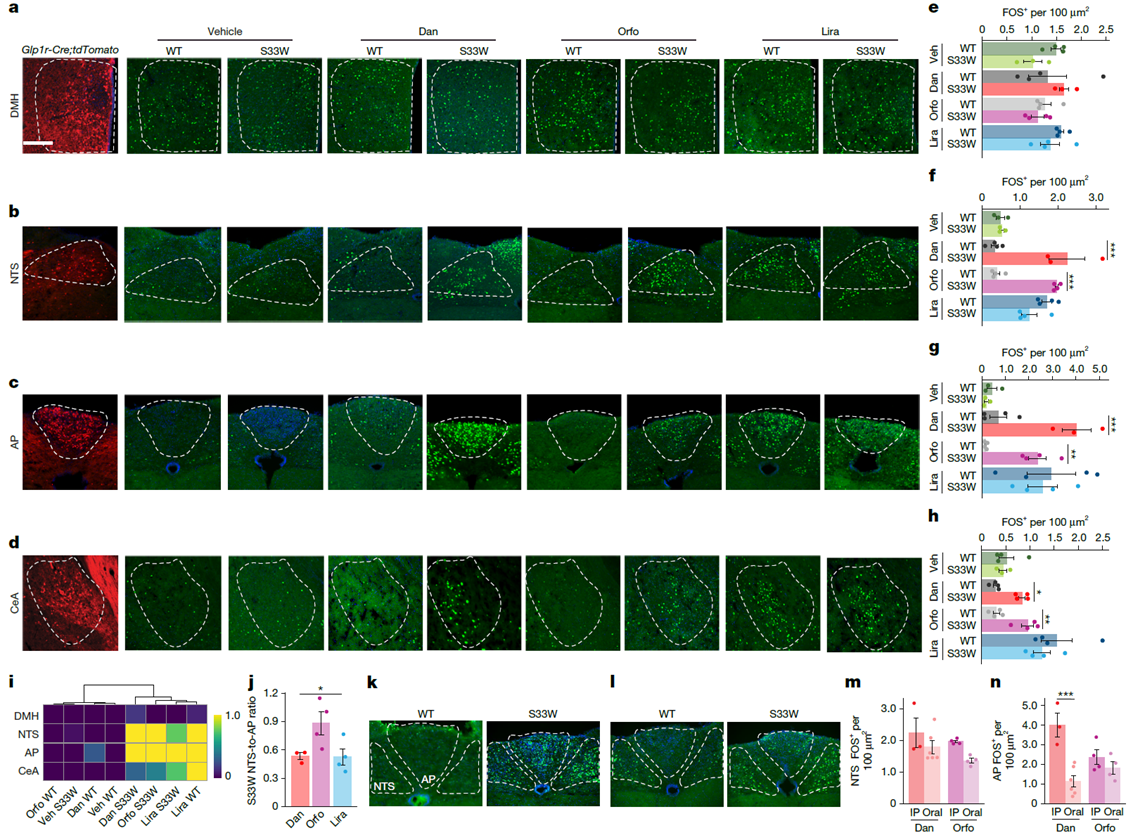
Figure 3. GLP-1RA activation in brain regions expressing GLP-1R
Specific Regulation of Hedonic Feeding by CeA Neurons
To clarify the function of specific brain regions, researchers expressed human GLP-1R in the central amygdala (CeA) of Glp1r-Cre mice. In this model, the small-molecule agonist Danuglipron specifically suppressed intake of hedonic high-fat diet (HFD) while having no effect on homeostatic standard diet (SD) intake. Optogenetic experiments further confirmed that direct stimulation of Glp1r neurons in the CeA was sufficient to suppress hedonic feeding. In addition, specific knockout of the CeA receptor in Glp1rflox/flox mice significantly attenuated liraglutide’s suppression of high-fat diet intake, establishing CeA neurons as a critical node mediating GLP-1RA inhibition of reward-driven feeding.
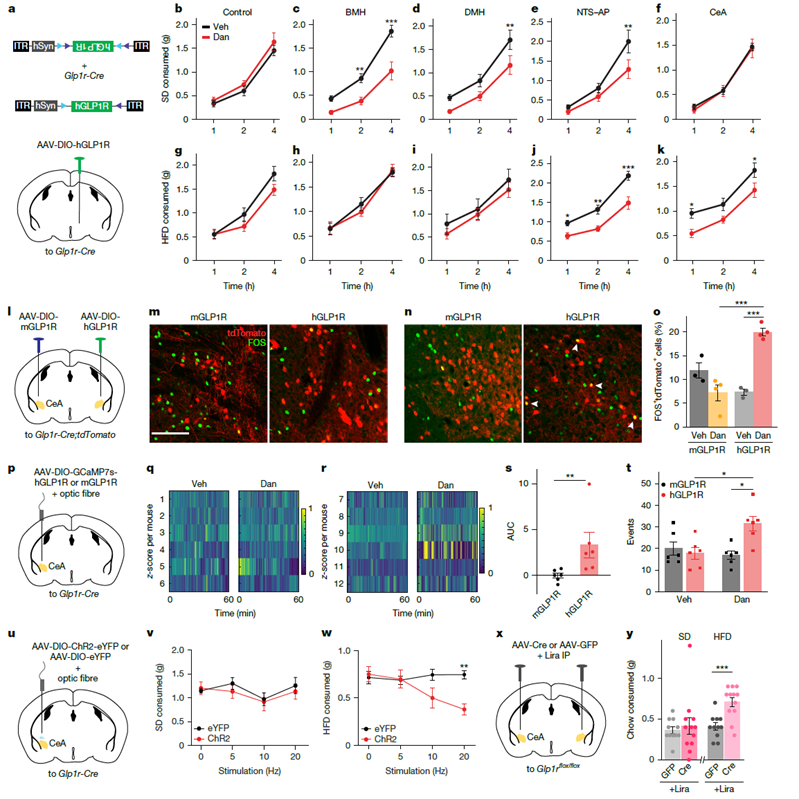
Figure 4. GLP-1 receptor activation in the central amygdala (CeA) suppresses hedonic food intake
Regulation of Dopamine Release via the CeA-VTA Pathway
Anatomical and functional experiments revealed the connectivity logic between Glp1r neurons in the CeA and the midbrain limbic reward system. Tracing experiments showed that CeA neurons provide direct synaptic input to dopaminergic neurons in the ventral tegmental area (VTA). In vivo recordings using the dLight1.3b fluorescent sensor demonstrated that small-molecule agonists effectively suppressed dopamine release peaks in the nucleus accumbens (NAc) during hedonic feeding. These findings prove that GLP-1R agonists reduce the neural reward value of hedonic feeding by activating CeA neurons and, in turn, the CeA-VTA pathway.
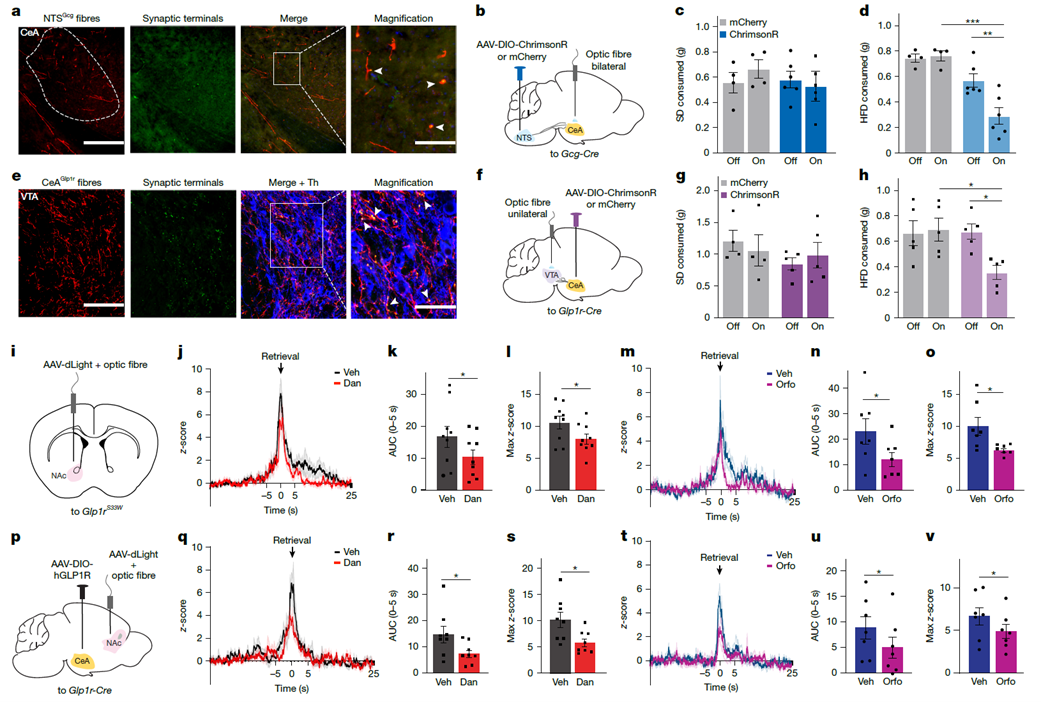
Figure 5. CeA/Glp1r neurons project to the VTA to modulate dopamine output to the NAc in response to high-fat diet (HFD)
Research Summary: This study identifies a neural pathway driven by the hindbrain nucleus tractus solitarius (NTS) and converging on the central amygdala (CeA). This pathway precisely suppresses reward-driven feeding behavior by modulating the mesolimbic dopamine system. The findings provide a new neurobiological explanation for how GLP-1R agonists improve metabolism and suggest potential clinical value for treating reward-system disorders such as substance use disorders and binge-eating disorder. The study emphasizes the importance of considering regulation of hedonic circuits when developing next-generation weight-loss drugs, enabling effective weight reduction while optimizing patient treatment experience.
abinScience GLP-1R & Metabolic Neuroscience Research Products
For the GLP-1R receptor studies, dopamine regulation, and hedonic feeding circuit analyses involved in the above research, abinScience offers a variety of reference antibodies targeting GLP-1R and GIPR, as well as detection kits and specific antibodies for agonists such as Orforglipron and Semaglutide, to support your metabolic and neuroscience research.
| Catalog No. |
Product Name |
| SW328036 |
Research Grade Orforglipron |
| HF737016 |
Research Grade Efpegerglucagon |
| HW328026 |
Research Grade Gulgafafusp Alfa |
| HW328016 |
Anti-Human GLP-1R Reference Antibody (GMA105, RUO) |
| HW595026 |
Anti-Human GIPR&GLP-1R Reference Antibody (AMG 133, RUO) |
| HW328036 |
Anti-Human GLP-1R Reference Antibody (mAb-36986, RUO) |
| HP899014 |
Anti-Semaglutide (GLP-1 analogue) Polyclonal Antibody |
| HP899013 |
Anti-Semaglutide (GLP-1 analogue) Recombinant Antibody (SAA2594) |
| HP899015 |
Anti-Semaglutide (GLP-1 analogue) Monoclonal Antibody (1A723) |
| DW328048 |
Orforglipron ELISA Kit |
References
This article is based on the latest research published in Nature. For detailed mechanisms and experimental data, please refer to the original paper.
- Godschall, E.N., Gungul, T.B., Sajonia, I.R. et al. A brain reward circuit inhibited by next-generation weight-loss drugs in mice. Nature (2026). https://doi.org/10.1038/s41586-026-10444-4

 中文
中文 English
English 한국어
한국어 日本語
日本語 Español
Español Français
Français Русский
Русский







