Pathogenesis and Precision Treatment Advances in Immune Thrombocytopenia (ITP)
Immune Thrombocytopenia (ITP), formerly known as “idiopathic thrombocytopenic purpura,” is an autoimmune disorder caused by acquired immune dysregulation. It primarily manifests as mucocutaneous bleeding, with severe cases potentially leading to intracranial hemorrhage. According to the latest clinical consensus, ITP is defined as a highly heterogeneous syndrome rather than a single disease. Epidemiologically, with more precise diagnostic techniques, the incidence of adult ITP shows a clear bimodal distribution across genders and age groups, suggesting that different immune pathogenic drivers may predominate at different ages.
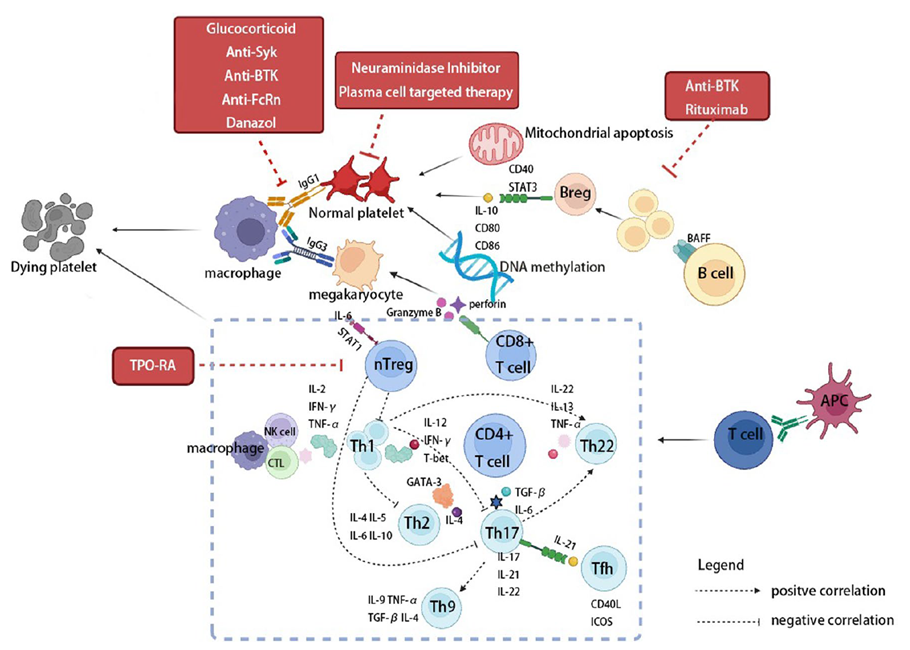
Figure 1. Simplified schematic of ITP pathogenesis and novel therapies
Pathogenic Mechanisms
The pathogenesis of ITP is complex and involves multiple pathways, including humoral immunity, cellular immunity, and megakaryocyte dysfunction. Patients produce IgG autoantibodies targeting platelet glycoproteins. These antibodies promote platelet clearance by splenic and hepatic macrophages through Fcγ receptor-mediated phagocytosis. They can also induce platelet desialylation, exposing N-acetylglucosamine residues that are recognized and cleared by the hepatic Ashwell-Morell receptor. Additionally, autoantibodies can bind to bone marrow megakaryocytes, inhibiting their maturation and platelet production, leading to reduced platelet generation. On the cellular immunity side, regulatory T cells (Treg) and regulatory B cells (Breg) are functionally defective, with Th1/Th17 subsets predominating. Cytotoxic CD8⁺ T cells directly induce apoptosis of platelets and megakaryocytes via the perforin/granzyme B pathway. Overexpression of B-cell activating factor (BAFF) further promotes the survival and antibody production of autoreactive B cells. Complement activation and epigenetic abnormalities (such as altered DNA methylation) also contribute, resulting in a dual pathology of “increased destruction + decreased production.”
Key Targets and Cutting-Edge Research Advances
In recent years, ITP treatment has entered a new era of precision immune modulation. In addition to traditional glucocorticoids, IVIG, rituximab, and thrombopoietin receptor agonists (TPO-RAs such as eltrombopag and romiplostim), several novel agents have entered clinical use or Phase III trials.
BTK Inhibitors: rilzabrutinib (Wayrilz)
BTK (Bruton’s tyrosine kinase) is a key kinase downstream of the B-cell receptor (BCR) and Fcγ receptor (FcγR) signaling pathways. In 2025, the oral reversible covalent BTK inhibitor rilzabrutinib (brand name Wayrilz) received FDA approval, becoming the first BTK inhibitor approved specifically for ITP. By selectively inhibiting BTK (via reversible covalent binding at the C481 site), the drug modulates immune responses through multiple pathways: it blocks BCR downstream signaling to inhibit autoreactive B-cell activation and autoantibody production; it also blocks FcγR-mediated macrophage phagocytosis, reducing clearance of antibody-coated platelets; meanwhile, it preserves platelet function mediated by G-protein-coupled receptors (such as CLEC-2), thereby avoiding increased bleeding risk. The LUNA 3 Phase III trial demonstrated that rilzabrutinib achieved rapid and sustained platelet increases in chronic ITP patients who had failed prior therapies, with significantly higher response rates than placebo and a favorable safety profile.
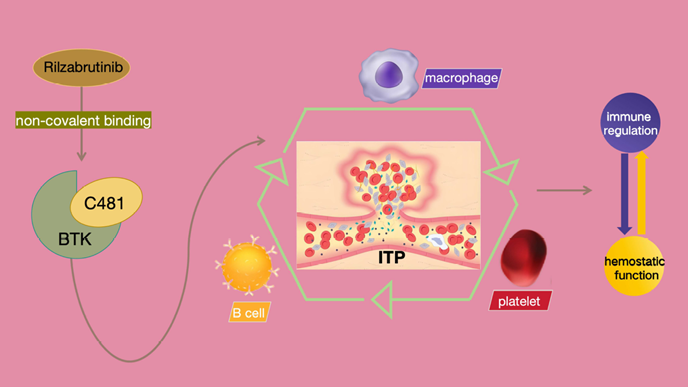
Figure 2. Mechanism of action and clinical translation of rilzabrutinib in ITP
FcRn Antagonists: efgartigimod
FcRn (neonatal Fc receptor) is a critical regulator of IgG homeostasis, protecting IgG from lysosomal degradation by binding it in acidic endosomes. The FcRn antagonist efgartigimod (an engineered IgG1 Fc fragment) competitively binds FcRn with high affinity, blocking IgG (including pathogenic autoantibodies) recycling and promoting its degradation, thereby specifically reducing circulating total IgG levels (typically by 60–80%). The ADVANCE IV Phase III trial showed that efgartigimod significantly increased the proportion of chronic ITP patients achieving sustained platelet responses (≥50×10⁹/L on ≥4 out of 6 scheduled visits), improved bleeding symptoms, and enhanced quality of life. Long-term efficacy is currently being evaluated in open-label extension studies.
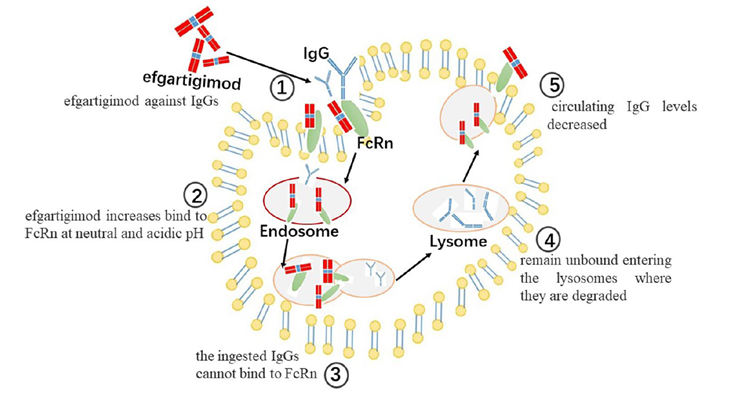
Figure 3. Schematic of efgartigimod’s pharmacological action
Anti-CD38 Monoclonal Antibodies
CD38 is highly expressed on long-lived plasma cells that produce autoantibodies. Anti-CD38 monoclonal antibodies (such as mezagitamab, CM313, and daratumumab) specifically deplete plasma cells through antibody-dependent cellular cytotoxicity (ADCC), complement-dependent cytotoxicity (CDC), and direct apoptosis induction, thereby reducing antibody production at its source. Phase II randomized controlled trials of CM313 and daratumumab, as well as the mezagitamab Phase II study, demonstrated rapid platelet count recovery and sustained responses in patients with refractory ITP. Phase III studies are currently underway.
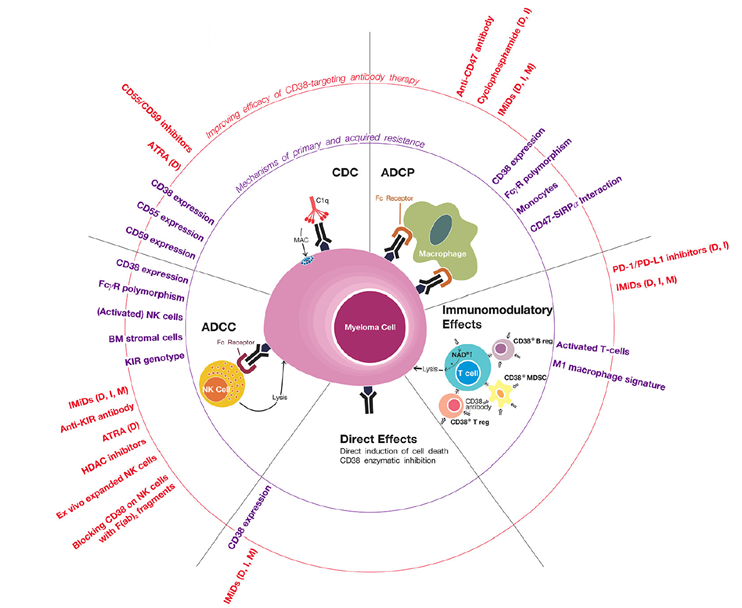
Figure 4. Mechanisms of primary and acquired resistance to anti-CD38 antibodies
Syk Inhibitors: fostamatinib (Tavalisse)
Syk (spleen tyrosine kinase) is a key upstream node in the FcγR signaling pathway. The Syk inhibitor fostamatinib (brand name Tavalisse, FDA-approved in 2018) blocks macrophage phagocytosis of IgG-coated platelets and inhibits B-cell activation and antibody production by inhibiting Syk. The FIT clinical trial program confirmed its ability to significantly increase platelet response rates in chronic ITP.
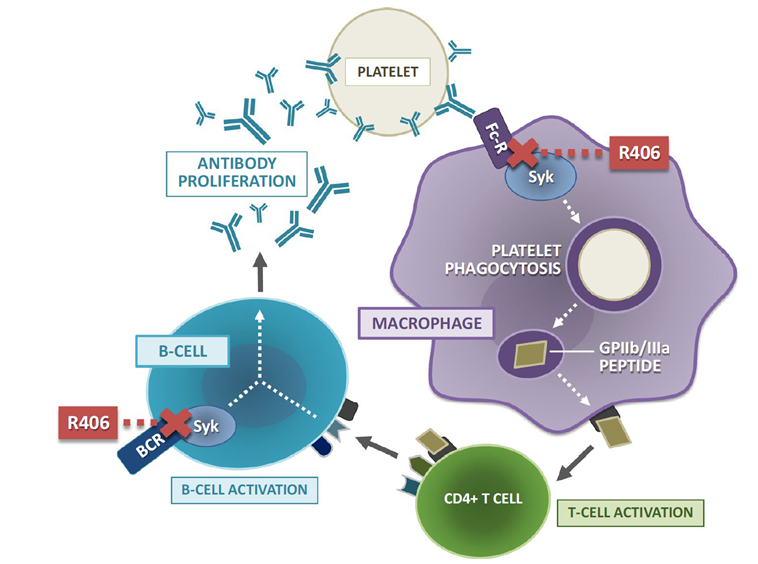
Figure 5. Mechanism of action of fostamatinib’s active metabolite R406 in immune thrombocytopenia
BAFF-R Inhibitors: ianalumab (VAY736)
BAFF-R (B-cell activating factor receptor) regulates B-cell survival, maturation, and proliferation. The BAFF-R inhibitor ianalumab (VAY736, a fully human IgG1 monoclonal antibody) inhibits autoreactive B-cell function by blocking BAFF-R signaling and enhancing ADCC-mediated B-cell depletion. Results from the VAYHIT2 Phase III trial showed that ianalumab combined with eltrombopag significantly prolonged time to treatment failure (TTF) and improved the 6-month stable response rate compared with placebo plus eltrombopag in primary ITP patients who failed first-line glucocorticoids. It also helped reduce eltrombopag dosage and may have potential disease-modifying effects.
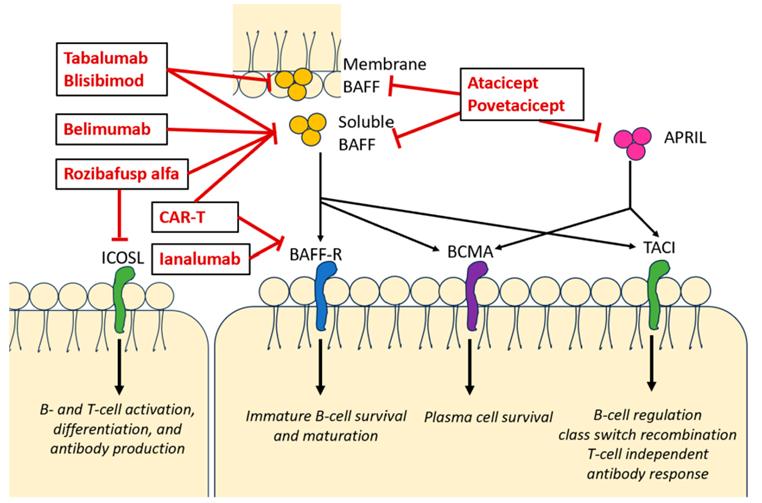
Figure 6. Mechanism of action of anti-BAFF agents
These precision targeted therapies together form the framework for a new era in ITP treatment. In the future, multi-target combinations or individualized regimens based on immune phenotypes are expected to further improve patient outcomes. In clinical practice, attention must be paid to each drug’s indications, monitoring parameters, and safety profiles.
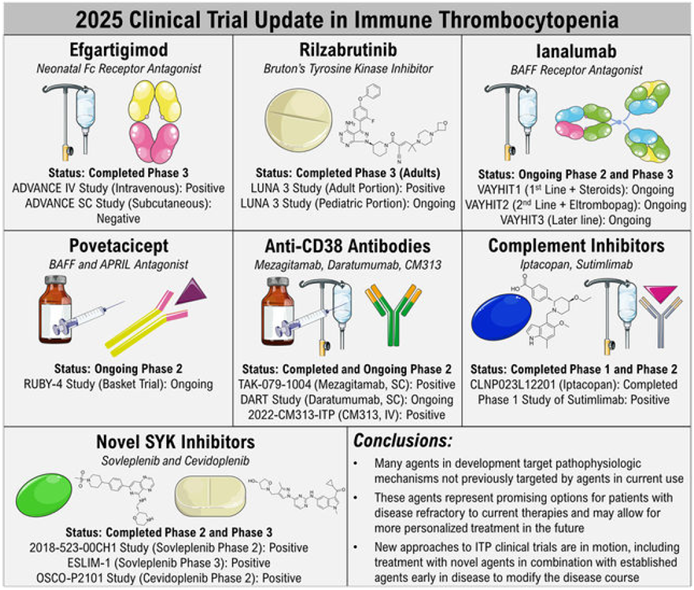
Figure 7. 2025 clinical trial updates in immune thrombocytopenia
abinScience Related Products
Below are abinScience’s latest recombinant proteins and antibodies targeting Immune Thrombocytopenia-related pathways. Catalog numbers are clickable links to the product pages.
Protein
| Catalog No. |
Product name |
| HT068012 |
Recombinant Human PIK3CD Protein, N-His |
| HF996011 |
Recombinant Human CD25/IL2RA Protein, C-His |
| HY477012 |
Recombinant Human CD42b/GP1BA Protein, N-His |
| HY386022 |
Recombinant Human CD16a/FCGR3A Protein, N-His |
| HY598012 |
Recombinant Human C1S Protein, N-His |
| HY598011 |
Recombinant Human C1S Protein, C-His |
| HY257012 |
Recombinant Human CD20/MS4A1 Protein, N-His |
| HY139012 |
Recombinant Human CD32/FCGR2A Protein, N-His |
| HB990012 |
Recombinant Human CD119/IFNGR1 Protein, N-His |
| HB011012 |
Recombinant Human CD22 Protein, N-His |
| HX061012 |
Recombinant Human FCGRT Protein, N-His |
| HC260012 |
Recombinant Human BTK Protein, N-His |
| HV212012 |
Recombinant Human CD257/TNFSF13B Protein, N-His |
| HW747012 |
Recombinant Human SYK Protein, N-His |
| HW601011 |
Recombinant Human CD110/MPL Protein, C-His |
| HB199012 |
Recombinant Human CD154/CD40LG/TNFSF5 Protein, C-His |
View remaining Protein products
Antibody
| Catalog No. |
Product name |
| HX061016 |
Research Grade Orilanolimab |
| HX061026 |
Research Grade Rozanolixizumab |
| HX061086 |
Research Grade imeroprubart |
| HV212026 |
Research Grade Tabalumab |
| HV212016 |
Research Grade Belimumab |
| HB199016 |
Research Grade Letolizumab |
| HB199026 |
Research Grade Dapirolizumab |
| HB199096 |
Research Grade Tegoprubart |
| HY598016 |
Research Grade Sutimlimab |
| HY598026 |
Research Grade Riliprubart |
| HY598036 |
Research Grade claseprubart |
| HY257036 |
Research Grade Ibritumomab |
| HY257466 |
Research Grade Ofatumumab |
| HY257026 |
Research Grade Ocrelizumab |
| HF996046 |
Research Grade Inolimomab |
| HF996036 |
Research Grade Camidanlumab |
| HF996136 |
Research Grade Vopikitug |
| HB011026 |
Research Grade Inotuzumab |
| HB011016 |
Research Grade Moxetumomab |
| HF996146 |
Research Grade imneskibart |
| HY386020 |
InVivoMAb Anti-Human CD16a/FCGR3A Antibody (CLB-Gran1) |
| HY598010 |
InVivoMAb Anti-Human C1S Antibody (TNT003) |
| MF996010 |
InVivoMAb Anti-Mouse CD25/IL2RA Antibody (PC61/PC61.5.3) |
| HW601010 |
InVivoMAb Anti-Human CD110/TPOR Antibody (3D9) |
| HX061010 |
InVivoMAb Anti-Human FCGRT/FCRN Antibody (Iv0203) |
| HY477107 |
Anti-Human CD42b/GP1BA Antibody (5G6) |
| HY386013 |
Anti-Human CD16a/FCGR3A Nanobody (SAA2051) |
| HY257107 |
Anti-Human CD20/MS4A1 Antibody (SAA0006) |
| HY257013 |
Anti-Human CD20/MS4A1 Nanobody (SAA1332) |
| HY139013 |
Anti-Human CD32/FcgR2 Nanobody (SAA2150) |
| HB990107 |
Anti-Human CD119/IFNGR1 Antibody (SAA0044) |
| HB199207 |
Anti-Human CD154/CD40LG/TNFSF5 Antibody (5C8) |
| HW601207 |
Anti-Human CD110/MPL Antibody (PHA38) |
| HW747013 |
Anti-Human SYK Antibody (SAA1636) |
| HX061227 |
Anti-Human FCGRT/FCRN Antibody (SAA0136), PE |
| HV212227 |
Anti-Human CD257/BAFF/TNFSF13B Antibody (SAA0081), PE |
View remaining Antibody products
Kit
References:
1. Cheekati M, Murakhovskaya I. Anti-B-Cell-Activating Factor (BAFF) Therapy: A Novel Addition to Autoimmune Disease Management and Potential for Immunomodulatory Therapy in Warm Autoimmune Hemolytic Anemia. Biomedicines. 2024 Jul 18;12(7):1597. doi: 10.3390/biomedicines12071597 . PMID: 39062171; PMCID: PMC11275058.
2. Neunert CE, Arnold DM, Grace RF, Kuhne T, McCrae KR, Terrell DR. The 2022 review of the 2019 American Society of Hematology guidelines on immune thrombocytopenia. Blood Adv. 2024 Jul 9;8(13):3578-3582. doi: 10.1182/bloodadvances.2023012541 . PMID: 38608258; PMCID: PMC11319830.
3. Liu L, Xiao Y, Jia Y, Shao Z, Shi J, Cui C. From C481 Resistance Evasion to Platelet Preservation: Rilzabrutinib Redefines ITP Targeted Therapy. Drug Des Devel Ther. 2025 Sep 11;19:8161-8180. doi: 10.2147/DDDT.S543620 . PMID: 40959738; PMCID: PMC12435525.
4. Yang Y, Shen Z, Shi F, Wang F, Wen N. Efgartigimod as a novel FcRn inhibitor for autoimmune disease. Neurol Sci. 2024 Sep;45(9):4229-4241. doi: 10.1007/s10072-024-07460-5 . Epub 2024 Apr 22. PMID: 38644454.
5. van de Donk NWCJ and Usmani SZ (2018) CD38 Antibodies in Multiple Myeloma: Mechanisms of Action and Modes of Resistance. Front. Immunol. 9:2134. doi: 10.3389/fimmu.2018.02134
6. Paik J. Fostamatinib: A Review in Chronic Immune Thrombocytopenia. Drugs. 2021 Jun;81(8):935-943. doi: 10.1007/s40265-021-01524-y . Epub 2021 May 10. Erratum in: Drugs. 2021 Jul;81(11):1361. doi: 10.1007/s40265-021-01540-y. PMID: 33970459.
7. Mititelu A, Onisâi MC, Roșca A, Vlădăreanu AM. Current Understanding of Immune Thrombocytopenia: A Review of Pathogenesis and Treatment Options. Int J Mol Sci. 2024 Feb 10;25(4):2163. doi: 10.3390/ijms25042163 . PMID: 38396839; PMCID: PMC10889445.
8. Zufferey A, Kapur R, Semple JW. Pathogenesis and Therapeutic Mechanisms in Immune Thrombocytopenia (ITP). J Clin Med. 2017 Feb 9;6(2):16. doi: 10.3390/jcm6020016 . PMID: 28208757; PMCID: PMC5332920.
9. Madkhali MA. Recent advances in the management of immune thrombocytopenic purpura (ITP): A comprehensive review. Medicine (Baltimore). 2024 Jan 19;103(3):e36936. doi: 10.1097/MD.0000000000036936 . PMID: 38241567; PMCID: PMC10798712.
10. Al-Samkari H. 2025 update on clinical trials in immune thrombocytopenia. Am J Hematol. 2024 Nov;99(11):2178-2190. doi: 10.1002/ajh.27448 . Epub 2024 Aug 6. PMID: 39105413; PMCID: PMC11469945.
11. Yan, X., Yun, Z., Tian, L. et al. Immune thrombocytopenia: a review of pathogenesis and current treatment. Discov Med 1, 57 (2024). https://doi.org/10.1007/s44337-024-00040-8
12. Dörner T, Posch MG, Li Y, Petricoul O, Cabanski M, Milojevic JM, Kamphausen E, Valentin MA, Simonett C, Mooney L, Hüser A, Gram H, Wagner FD, Oliver SJ. Treatment of primary Sjögren’s syndrome with ianalumab (VAY736) targeting B cells by BAFF receptor blockade coupled with enhanced, antibody-dependent cellular cytotoxicity. Ann Rheum Dis. 2019 May;78(5):641-647. doi: 10.1136/annrheumdis-2018-214720 . Epub 2019 Mar 2. PMID: 30826774.

 中文
中文 English
English 한국어
한국어 日本語
日本語 Español
Español Français
Français Русский
Русский









