Key Takeaways
A Wake-Up Call at Sea: In May 2026, a transnational cruise ship outbreak of Andes virus (ANDV)—the only hantavirus capable of limited human-to-human transmission—highlighted the pathogen's cross-border spillover risk.
Two Syndromes, One Vascular Target: Hantavirus infection causes either Hemorrhagic Fever with Renal Syndrome (HFRS) or Hantavirus Pulmonary Syndrome (HPS), with mortality up to 60%; both are driven by endothelial barrier disruption, a cytokine storm, and coagulation dysfunction.
No Specific Cure—Research Is the Defense: With no approved antiviral therapy, neutralizing antibodies against Gn and Gc remain the primary vaccine focus, and deep characterization of core viral proteins (NP, Gn, Gc) is critical for advancing diagnostics, vaccines, and therapeutics.
In May 2026, the World Health Organization (WHO) officially reported a transnational cluster of hantavirus infections: the Dutch polar expedition cruise ship Hondius reported 2 confirmed and 5 suspected hantavirus cases during its voyage, including 3 deaths and 1 critical case. The vessel is currently under full quarantine in the waters off Cape Verde, and health authorities across multiple countries have initiated contact tracing and emergency responses.
This rare maritime cluster outbreak has brought hantavirus—a long-neglected zoonotic pathogen—back into the spotlight of global scientific research. The continuous sporadic global prevalence of hantavirus, combined with its high pathogenicity and stealthy nature, presents ongoing challenges for pathogen surveillance, basic research, and epidemic control.
1. Core Pathogenic Characteristics of Hantavirus
According to the latest 2024 classification by the International Committee on Taxonomy of Viruses (ICTV), hantaviruses belong to the genus Orthohantavirus, family Hantaviridae, order Bunyavirales. They are enveloped, single-stranded negative-sense RNA viruses and represent one of the most widely distributed rodent-borne viruses globally.
1.1 Viral Morphology and Genomic Structure
Hantavirus virions are spherical or oval, with a diameter ranging from 78 to 210 nm (averaging approximately 120 nm). The nucleocapsid exhibits a helical symmetry, enveloped by a double-layered lipid bilayer. The viral envelope surface is covered with spike-like projections composed of glycoproteins.
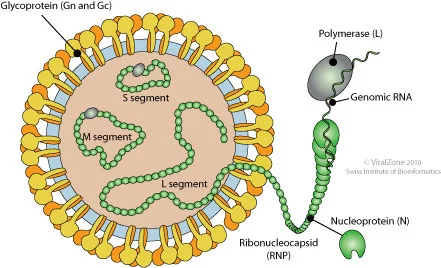
Figure 1. Structure of Hantavirus (Source: Viral Zone)
The viral genome spans approximately 10.5 to 14.6 kb and is divided into three distinct RNA segments: Large (L), Medium (M), and Small (S). These segments encode highly conserved core proteins, which serve as essential targets for viral classification, diagnostic detection, and functional research:
- L Segment: Encodes the RNA-dependent RNA polymerase (RdRp), which drives viral genome replication and transcription. As the core functional protein for viral proliferation, RdRp is a crucial target for the development of antiviral therapeutics.
- M Segment: Encodes the glycoprotein precursor (GPC), which is subsequently cleaved by host proteases into two envelope glycoproteins, Gn and Gc. These proteins primarily mediate viral attachment to host cell receptors and subsequent membrane fusion, and they act as the principal targets recognized by host neutralizing antibodies.
- S Segment: Encodes the nucleocapsid protein (NP), which encapsidates the viral genome to form the ribonucleoprotein (RNP) complex, maintaining genomic stability. Given its robust immunogenicity, NP is a critical target for serological diagnostics.
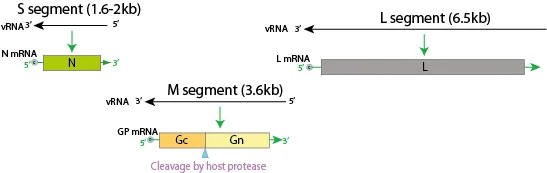
Figure 2. Genome of Hantavirus (Source: Viral Zone)
1.2 Major Epidemic Strains and Pathogenic Disparities
The genus Orthohantavirus encompasses over 50 viral species capable of infecting mammals. Based on geographic distribution, primary hosts, and clinical pathogenicity, they are generally classified into two major evolutionary lineages with significant differences in pathogenesis:
| Lineage |
Primary Endemic Region |
Representative Strains |
Associated Disease |
Case Fatality Rate |
| Old World Hantaviruses |
Eurasia (including China) |
Hantaan virus (HTNV), Seoul virus (SEOV), Puumala virus (PUUV) |
Hemorrhagic Fever with Renal Syndrome (HFRS) |
0.5%-15% |
| New World Hantaviruses |
The Americas |
Sin Nombre virus (SNV), Andes virus (ANDV) |
Hantavirus Pulmonary Syndrome (HPS) |
30%-60% |
In this recent cruise ship outbreak, the strain responsible for the severe and fatal cases was identified as the Andes virus (ANDV). Notably, ANDV is the only hantavirus strain documented to undergo limited human-to-human transmission. Its primary reservoir is the long-tailed pygmy rice rat, native to southern South America; the port of Ushuaia in Argentina (the origin of the cruise) is a known endemic hotspot for this specific strain.
2. Transmission Routes of Hantavirus
Hantaviruses are archetypal host-restricted viruses. Natural infection does not cause clinical disease in the rodent reservoir; rather, it results in a persistent, lifelong infection where the virus is continuously shed through saliva, urine, and feces, thereby establishing natural foci of infection. The primary routes of human infection include:
Aerosol Inhalation
Inhaling aerosols contaminated by the excreta of infected rodents is the predominant transmission route. It poses the highest risk for individuals engaged in fieldwork or exposed to confined environments.
Contact Transmission
Direct contact with the excreta or secretions of infected animals, or sustaining a bite from a carrier animal, allows the virus to breach compromised skin or mucous membranes.
Gastrointestinal Transmission
Consumption of food or water contaminated by infected animal excreta.
Human-to-Human Transmission
This is an extreme rarity strictly associated with ANDV infections, where limited transmission can occur in settings involving exceedingly close contact. Routine social interactions present no risk. Epidemiological investigations into the recent cruise ship outbreak strongly suggest this atypical mode of transmission.
Figure 3. Transmission Routes of Hantavirus (DOI: 10.1111/1469-0691.12291)
3. Molecular Mechanisms of Hantavirus Pathogenesis
The fundamental pathogenic basis of hantavirus infection involves vascular endothelial cell injury, abnormal increases in vascular permeability, coagulation cascade dysregulation, and host immunopathological damage. The virus exhibits a pantropic affinity for systemic vascular endothelial cells. By binding its envelope glycoproteins to host cell surface β3 integrin receptors, the virus internalizes, replicates within monocytes and endothelial cells, and disseminates to multiple organ systems via the bloodstream.
1. Mechanisms of Vascular Endothelial Barrier Dysfunction
Disruption of the vascular endothelial barrier serves as the common pathological foundation across all clinical phenotypes of hantavirus infection. Even without inducing direct cytolysis, the virus compromises barrier integrity through multiple pathways:
- Direct Disruption of Intercellular Junctions: The viral Gn protein can directly bind to the extracellular domain of VE-cadherin (a core protein in endothelial adherens junctions), destabilizing its homodimeric structure. Concurrently, by activating host signaling cascades, the virus induces the phosphorylation, endocytosis, and degradation of tight junction proteins (occludin, claudin-5) and VE-cadherin, physically compromising endothelial connections.
- Cytoskeletal Rearrangement: Viral infection activates the Rho GTPase signaling pathway, triggering actin cytoskeletal remodeling and stress fiber formation. This leads to endothelial cell contraction, widening of intercellular gaps, and an abnormal elevation in vascular permeability. Plasma leakage into the alveoli precipitates lethal respiratory distress, whereas leakage into the kidneys causes renal interstitial edema and tubular necrosis—underpinning the core pathological hallmarks of HPS and HFRS, respectively.
2. Mechanisms of Immunopathological Damage
The NSs, NP, and Gn proteins encoded by hantaviruses inherently antagonize the host's innate immune responses, delaying the onset of interferon signaling and facilitating robust viral replication early in infection. In the later stages, the accumulation of viral nucleic acids and proteins triggers a hyperactivation of the immune system that culminates in a cytokine storm, further exacerbating vascular endothelial injury.
- Innate Immune Hyperactivation: Viral RNA is recognized by pattern recognition receptors (PRRs) such as TLR3/7/8 and RIG-I/MDA5, activating the NF-κB and IRF3/7 signaling pathways. This induces massive production of pro-inflammatory cytokines (IL-6, TNF-α, IL-1β), chemokines, and type I interferons. Simultaneously, the activation of the NLRP3 inflammasome promotes the maturation and secretion of IL-1β and IL-18, triggering a cascading inflammatory response.
- The Dual Role of Adaptive Immunity: While virus-specific CD8+ T cells are crucial for viral clearance, overactivated cytotoxic T lymphocytes can directly mediate the lysis of infected endothelial cells while releasing copious pro-inflammatory factors, exacerbating immunopathological damage. Conversely, neutralizing antibodies targeting Gn/Gc effectively block viral entry. This is the only validated protective immune response and remains the primary focus for vaccine development.
3. Coagulopathy and Hemorrhagic Mechanisms
The hemorrhagic phenotype stems from two core mechanisms: the disruption of vascular endothelial barrier integrity, and profound platelet dysfunction coupled with coagulation system dysregulation. By binding directly to platelets via β3 integrins, the virus inhibits platelet activation, aggregation, and adhesion, leading to thrombocytopenia and bleeding diathesis. Furthermore, the hyperinflammatory response triggers excessive activation of the coagulation cascade, precipitating disseminated intravascular coagulation (DIC). The rapid consumption of coagulation factors and platelets aggravates hemorrhage and multi-organ damage.
4. Clinical Features of Hantavirus Infection
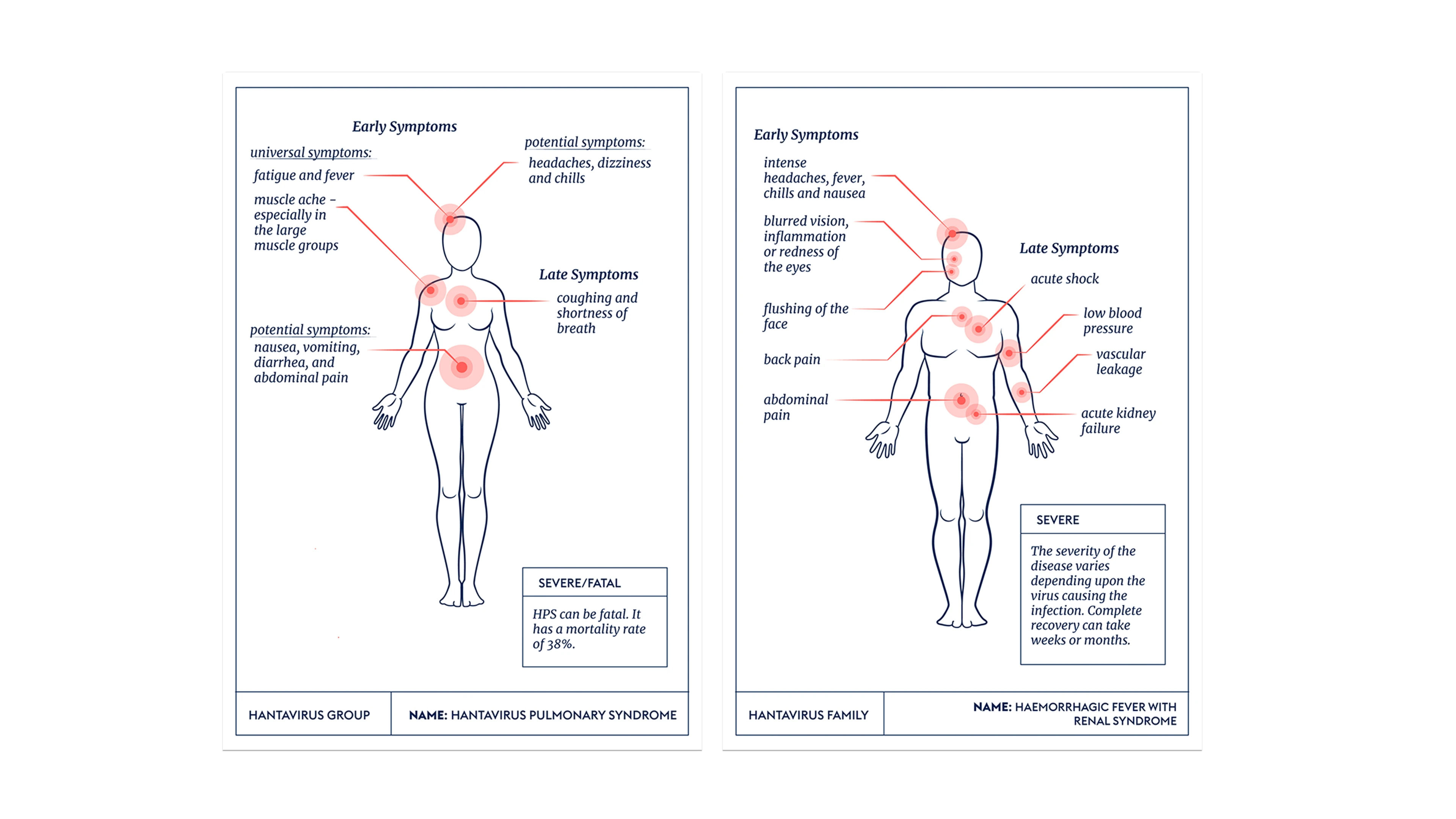
Figure 4. Clinical Features of Hantavirus (Source: CEPI. https://cepi.net/hantaviruses)
Depending on the infecting strain, the clinical presentation primarily manifests as one of two distinct syndromes:
Hemorrhagic Fever with Renal Syndrome (HFRS)
The incubation period typically ranges from 7 to 14 days. The classic clinical course progresses through five stages: febrile, hypotensive shock, oliguric, polyuric, and convalescent phases. The core clinical triad consists of "fever, hemorrhage, and renal damage." Typical cases present with characteristic features such as the "three pains" (headache, lower back pain, orbital pain) and the "three rednesses" (flushing of the face, neck, and upper chest). In severe cases, mortality arises from acute renal failure, hypovolemic shock, or DIC. The central pathological mechanisms involve direct virally-mediated injury and Type III hypersensitivity reactions driven by immune complexes, which induce massive inflammatory cytokine release and subsequent multi-organ damage.
Hantavirus Pulmonary Syndrome (HPS)
The incubation period spans 1 to 5 weeks. The prodrome presents with non-specific flu-like symptoms, which within 3 to 7 days rapidly escalate into Acute Respiratory Distress Syndrome (ARDS). Massive capillary leakage floods the alveoli with serous fluid, causing progressive dyspnea and severe hypoxemia. The condition can swiftly deteriorate into respiratory failure and shock, with a mortality rate of approximately 40%. The fatal cases documented in the recent cruise ship outbreak align precisely with the severe disease progression of HPS. The primary pathogenic driver is a virally triggered "cytokine storm" (e.g., IL-6 pathway hyperactivation) that severely compromises the pulmonary vascular endothelial barrier, rather than direct lytic destruction of lung tissue by the virus. This mechanism is the root cause of the rapid disease progression and the profound difficulty in clinical intervention.
As a globally distributed zoonotic pathogen, hantavirus remains a significant global public health threat due to its continuous sporadic outbreaks and sudden cluster events. The recent transnational cruise ship outbreak serves as a stark reminder that the risks of cross-regional and cross-species viral transmission are ever-present. Breakthroughs in foundational scientific research form the bedrock of our defense. An in-depth structural and functional characterization of core viral proteins—namely NP, Gn, and Gc—not only elucidates the underlying biological nature and pathogenic mechanisms of the virus but also provides crucial support for the development of novel diagnostics, vaccines, and antiviral therapeutics.
5. abinScience Products for Hantavirus Research
abinScience offers high-quality recombinant proteins and highly specific antibodies tailored for hantavirus research, supporting diverse applications in viral pathology and related immune pathway investigations.
| Types of virus |
Product Name |
Catalog No. |
| Araucaria virus |
Araucaria virus Nucleoprotein Recombinant Protein (N-His) |
VK132012 |
| Anti-Araucaria virus Nucleoprotein Polyclonal Antibody |
VK132014 |
| BCCV |
Anti-BCCV N/Nucleoprotein Polyclonal Antibody |
VK484014 |
| BCCV N/Nucleoprotein Recombinant Protein (N-His) |
VK484012 |
| Anti-BCCV Gc/Glycoprotein C Polyclonal Antibody |
VK646014 |
| Anti-BCCV Gn/Glycoprotein N Polyclonal Antibody |
VK646024 |
| BCCV Gc/Glycoprotein C Recombinant Protein (N-His) |
VK646012 |
| BCCV Gn/Glycoprotein N Recombinant Protein (N-His) |
VK646022 |
| BCCV Glycoprotein N/Gn Recombinant Protein (C-Fc) |
VK646051 |
| HTNV |
InVivoMAb Anti-Hantaan virus Glycoprotein C/Gc Broad-Neutralizing Antibody (Iv0260) |
VK646020 |
| InVivoMAb Anti-Hantaan virus Glycoprotein C/Gc Broad-Neutralizing Antibody (Iv0261) |
VK646030 |
| Anti-Hantaan virus/HTNV GP Antibody (3G1) |
VK646013 |
| Anti-Hantaan virus/HTNV GP Antibody (A5) |
VK646023 |
| Anti-Hantaan virus/HTNV Glycoprotein N/Gn Antibody (AH100) |
VK646033 |
| Anti-Hantaan virus/HTNV Glycoprotein N/Gn Antibody (SAA2516) |
VK646043 |
| HTNV/KHFV Glycoprotein N/Gn/G1 Recombinant Protein (C-His) |
VK646061 |
| HTNV/KHFV Glycoprotein N/Gn/G1 Recombinant Protein (C-Fc) |
VK646071 |
| Anti-Orthohantavirus puumalaense Glycoprotein C/Gc Polyclonal Antibody |
VK646034 |
| PUUV |
Anti-Orthohantavirus puumalaense GP Reference Antibody (ADI-42898, RUO) |
VK646016 |
| Anti-Orthohantavirus puumalaense GP Reference Antibody (ADI-65533, RUO) |
VK646026 |
| Anti-Orthohantavirus puumalaense GP Reference Antibody (ADI-65534, RUO) |
VK646036 |
| InVivoMAb Anti-Orthohantavirus puumalaense GP/Envelopment polyprotein Antibody (4G2) |
VK646010 |
| Anti-Orthohantavirus puumalaense Glycoprotein N/Gn Polyclonal Antibody |
VK646044 |
| Orthohantavirus puumalaense Glycoprotein C/Gc Recombinant Protein (C-Fc) |
VK646011 |
| Orthohantavirus puumalaense Glycoprotein C/Gc Recombinant Protein (C-His) |
VK646021 |
| Orthohantavirus puumalaense Glycoprotein C/Gc Recombinant Protein (N-His) |
VK646032 |
| Orthohantavirus puumalaense Glycoprotein N/Gn Recombinant Protein (C-Fc) |
VK646031 |
| Orthohantavirus puumalaense Glycoprotein N/Gn Recombinant Protein (C-His) |
VK646041 |
| Orthohantavirus puumalaense Glycoprotein N/Gn Recombinant Protein (N-His) |
VK646042 |
| Anti-SNV Glycoprotein Gn Polyclonal Antibody |
VK018014 |
| SNV |
Anti-SNV Glycoprotein Gn Antibody (SNV-42) |
VK018013 |
| SNV Glycoprotein Gn Recombinant Protein (C-Fc) |
VK018011 |
| SNV Glycoprotein Gn Recombinant Protein (N-His) |
VK018012 |
| Anti-SNV Glycoprotein Gc Polyclonal Antibody |
VK019014 |
| SNV Glycoprotein Gc Recombinant Protein (C-Strep) |
VK019011 |
| SNV Glycoprotein Gc Recombinant Protein (N-His) |
VK019012 |
References
[1] Afzal S, Ali L, Batool A, Afzal M, Kanwal N, Hassan M, Safdar M, Ahmad A, Yang J. Hantavirus: an overview and advancements in therapeutic approaches for infection. Front Microbiol. 2023 Oct 12;14:1233433. doi: 10.3389/fmicb.2023.1233433. Erratum in: Front Microbiol. 2023 Dec 12;14:1343080. doi: 10.3389/fmicb.2023.1343080. PMID: 37901807; PMCID: PMC10601933.
[2] Sehgal A, Mehta S, Sahay K, Martynova E, Rizvanov A, Baranwal M, Chandy S, Khaiboullina S, Kabwe E, Davidyuk Y. Hemorrhagic Fever with Renal Syndrome in Asia: History, Pathogenesis, Diagnosis, Treatment, and Prevention. Viruses. 2023 Feb 18;15(2):561. doi: 10.3390/v15020561. PMID: 36851775; PMCID: PMC9966805.
[3] Vaheri A, Strandin T, Hepojoki J, Sironen T, Henttonen H, Mäkelä S, Mustonen J. Uncovering the mysteries of hantavirus infections. Nat Rev Microbiol. 2013 Aug;11(8):539-50. doi: 10.1038/nrmicro3066. PMID: 24020072.
[4] Avšič-Županc T, Saksida A, Korva M. Hantavirus infections. Clin Microbiol Infect. 2019 Apr;21S:e6-e16. doi: 10.1111/1469-0691.12291. Epub 2015 Jun 22. PMID: 24750436.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.

 中文
中文 English
English 한국어
한국어 日本語
日本語 Español
Español Français
Français Русский
Русский






