Challenges in Antigen-Specific T Cell Functional Assays
Ensuring the reliability and comparability of antigen-specific T cell functional assays has long been a challenge in both research and clinical trials for immune monitoring. Although traditional polychromatic flow cytometry has advanced in parameter multiplexing, its accuracy and cross-laboratory consistency are often limited. OMIP-009 introduces a distinctive 10 color panel design focused on the reliability of functional signals, ensuring comparability and interpretability across different laboratories and instruments.
Quantitative Consistency of Functional Signals
The design of OMIP-009 moves beyond the conventional approach of simply increasing parameters. By optimizing the configuration of fluorescence channels, it clearly prioritizes key cytokines (such as IFN γ, IL 2, and TNF α) as core readouts, ensuring that functional signals remain consistent across laboratories, batches, and instruments. This innovative strategy provides a standardized tool for evaluating antigen specific T cell function.
Innovative Experimental Design Enhances Data Reproducibility
OMIP-009 not only ensures reliable cytokine detection but also introduces strict controls over experimental consistency, including systematic optimization of staining order, internalization effects, and “escapee” phenomena. This approach represents a shift toward a more scientific and systematic practice in flow cytometry-based immune monitoring, advancing the field as a whole.
In immune monitoring using polychromatic flow cytometry, achieving reliable and comparable antigen-specific T‑cell functional assays remains a major hurdle in research and clinical studies. OMIP-009 proposes a unique 10‑color panel design that does not merely pursue parameter stacking but employs a systematic approach to ensure functional signals are comparable and interpretable across different laboratories.
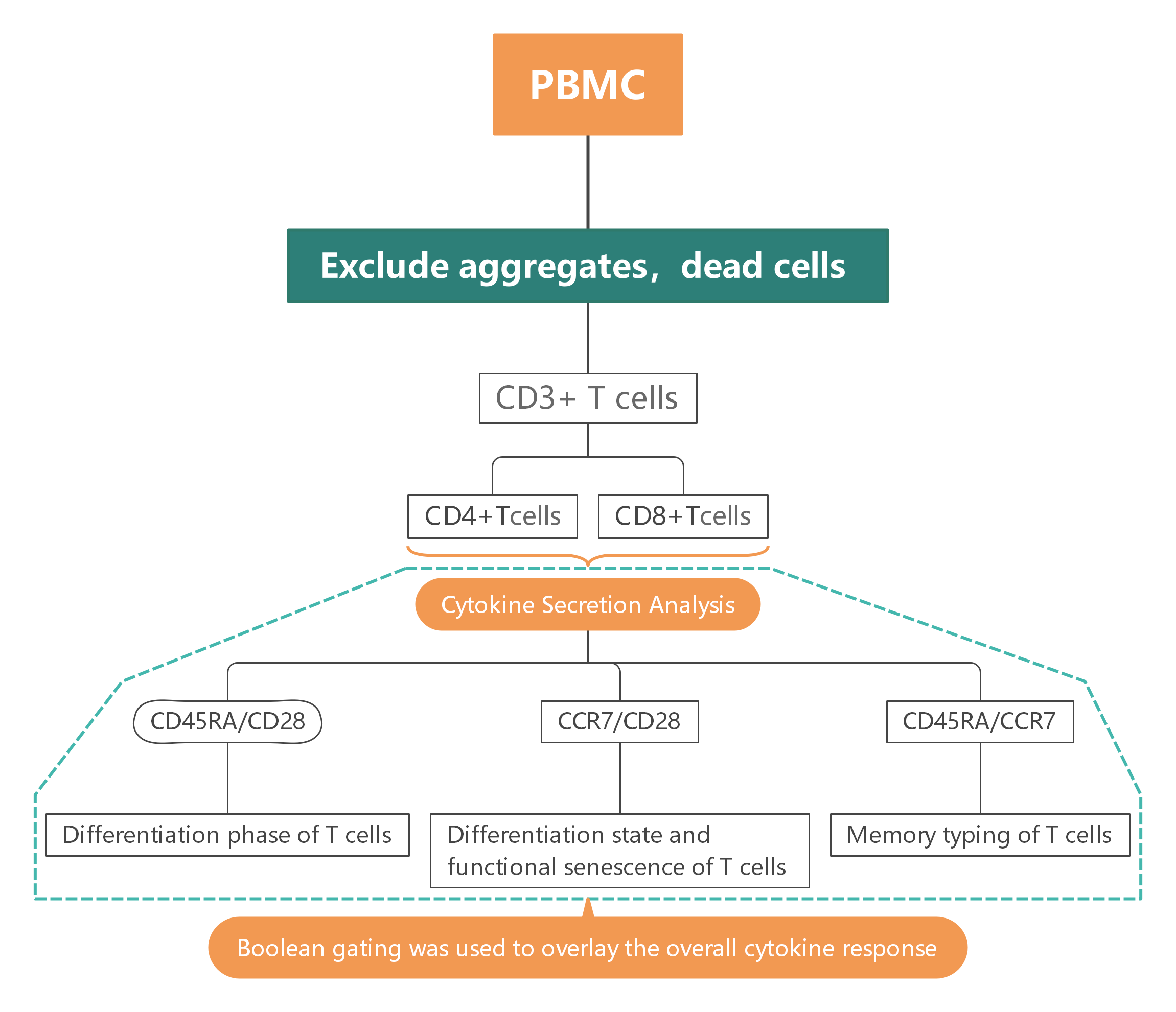
2. Gate Logic
1
T Cell Subpopulation Selection and Isolation
Exclude cell aggregates and dead cells, gate live CD3+ T cell populations, and use CD4 and CD8 to further differentiate CD4+ and CD8+ T cell subsets.
2
Cytokine Secretion Analysis
Use IFN-γ, IL-2, and TNF markers to analyze cytokine secretion profiles of different T cell subsets.
3
Multifunctional T Cell Population Analysis
Utilize Boolean gating to construct positive/negative combination matrices, identifying T cell populations co-expressing single, double, or multiple factors.
3. Experimental Results
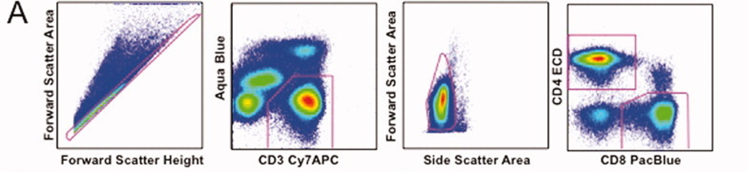
1). Exclude cell aggregates and dead cells, then identify live parent T cell populations using CD3 and Aqua. Refine the population further using FSC/SSC, followed by gating CD4 and CD8 to isolate specific T cell subsets for analysis.
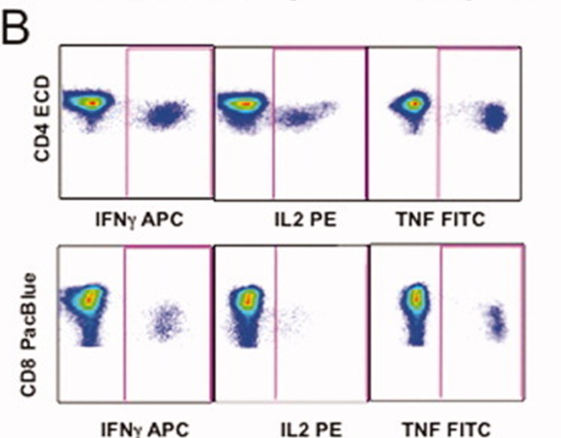
2). Cytokine secretion profiles of CD4+ and CD8+ T cell subsets were analyzed by gating for IFN-γ, IL-2, and TNF, revealing their functional responses.
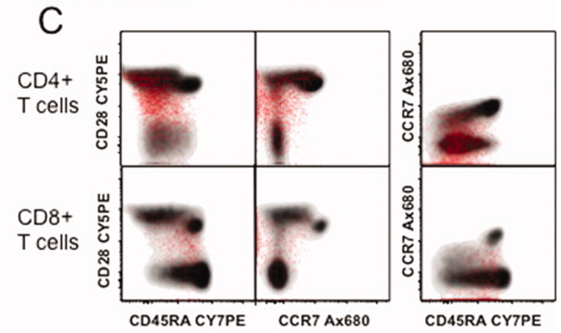
3). Naïve and memory T cell populations in both CD4+ and CD8+ subsets were assessed. Boolean gating was then used to overlay the overall cytokine response (shown in red) onto the corresponding CD4+ or CD8+ T cell lineage (shown in gray), enabling identification of activated and mature cell phenotypes.
4. Panel Interpretation
4.1 From "Multicolor Detection" to "Cross-Laboratory Comparable Functional Assays"
The major breakthrough of OMIP-009 lies in its shift from focusing on the number of parameters to prioritizing the quantitative consistency of functional signals. It explicitly places functional molecules such as IFNγ, IL-2, and TNF at the highest priority, and configures fluorescence channels and antibody combinations around these key indicators. The starting point is: real biological responses are often as low as 1% or even lower, so maintaining statistical reliability in the presence of noise is more important than increasing the number of markers.
As such, OMIP-009 is more like a "measurement system definition"—ensuring that regardless of laboratory, batch, or instrument, the functional signals measured have the same dimensionality and interpretability.
4.2 Rejecting Signal Amplification, Retaining True Distribution of Activation
In designing the stimulation system, OMIP-009 intentionally discarded the commonly used CD28/49d co-stimulation. Experimental validation showed that while co-stimulation could significantly increase the cytokine positivity rate, it simultaneously raised background noise and altered the true distribution pattern of responding cells.
OMIP-009 opted for direct stimulation of PBMCs with a peptide pool, capturing the T cell response to true antigens without introducing external signal amplification. This reflects a rare principled trade-off: it prioritizes the authenticity of the response, even if the signal is smaller, over the "artificially high" signal from amplified conditions. This "anti-activation" design philosophy is the fundamental reason OMIP-009 can be used for vaccine response analysis—it provides not maximal signal, but a true projection of response intensity under physiological conditions.
4.3 Integrating Staining Order, Endocytosis Effects, and the "Escapee" Phenomenon into Systematic Optimization
The optimization of the staining process in OMIP-009 is not merely about "dye testing," but a physical-level system correction of the experiment. Early tests revealed two main issues:
① Endocytosis of Surface Markers — After activation, CD4/CD8 signals weakened, leading to gating loss.
② "Escapee" Phenomenon — Low molecular weight dyes (e.g., Pacific Blue) would migrate to the high SSC region after centrifugation, causing erroneous exclusion.
In response, OMIP-009 converted CD4/CD8 staining to intracellular staining, fixed the cells before analysis, and systematically compared the staining order of viability dyes (AquaBlue) with CD3. This led to the establishment of a standardized workflow that balances signal stability with background control.
This optimization was not a mere "dye enhancement" but an acknowledgment that sample handling, washing, fixation, and staining order are experimental variables that must be designed, validated, and standardized. OMIP-009 thus became the first panel system to explicitly introduce "experimental step consistency" as a controlled variable. This concept was later extended to the quality control framework for all clinical OMIP panels.
5. Applications
Antigen-Specific T Cell Function Assessment T Cell Responses in Cancer Immunology T Cell Function Analysis in ImmunotherapyPreclinical vaccine research Cytokine Profiling of T Cells in Clinical Immunology Vaccine Immune Response Monitoring T Cell Function Monitoring in Autoimmune Diseases
6. Conclusion
By placing the reliability of functional signals, experimental system stability, and result reproducibility at its core, OMIP-009 embodies the transition of flow cytometry‑based immune detection from empirical operation to a scientific, systematic discipline. It not only provides a standardized, cross‑laboratory comparable protocol for evaluating antigen‑specific T‑cell function but also establishes a methodological foundation for the design of subsequent functional panels and clinical immune monitoring, marking a critical step forward in the scientific rigor and interpretability of polychromatic flow cytometry experiments.
Get OMIP-009 Compatible Flow Cytometry Antibodies
abinScience provides validated Flow Cytometry Antibodies covering key targets in this panel, supporting your Antigen-Specific T Cells research
References
[1] Lamoreaux L, Koup RA, Roederer M. OMIP-009: Characterization of antigen-specific human T-cells. Cytometry A. 2012 May;81(5):362-3. doi: 10.1002/cyto.a.22042. Epub 2012 Mar 21.

About Us
As a strategic venture of AtaGenix (established 2011), abinScience was founded in 2023 to deliver premium life science reagents that accelerate discovery. Our flow cytometry antibody products cover commonly used detection markers, with a wide variety to meet the research needs of multiple species (Human/Mouse/Rat/Dog/Hamster/Monkey, etc.). We provide stable and reliable support for scientific research.
Explore abinScience Flow Cytometry Antibodies

 中文
中文 English
English 한국어
한국어 日本語
日本語 Español
Español Français
Français Русский
Русский