Traumatic brain injury (TBI) affects approximately 69 million people worldwide each year, with mild TBI (mTBI) accounting for 80%–90% of cases. Repeated mild traumatic brain injury (rmTBI), a common subtype, can lead to persistent symptoms such as headache, depression, and memory deficits, and significantly increases the risk of neurodegenerative diseases including Parkinson’s disease and Alzheimer’s disease. Currently, the pathological mechanisms of rmTBI remain incompletely understood, and no specific clinical treatments are available. Ferroptosis is an iron-dependent form of programmed cell death characterized by accumulation of lipid peroxides and reactive oxygen species (ROS), displaying morphological similarities to neuronal damage observed in rmTBI. Naozhenning, a traditional Chinese herbal compound developed in 1976, has demonstrated clinical efficacy in brain injury-related conditions and has been shown in animal studies to improve mitochondrial function and protect myelin and axons. However, its active components and mechanism of action in rmTBI were previously unclear. Using a rat model of repeated mild cerebral concussion (MCC) combined with multiple experimental techniques, this study systematically investigated the role of ferroptosis in rmTBI and the neuroprotective mechanism of Naozhenning.
Impact Height Influences Cognitive Function and Neuronal Damage in the MCC Model
The MCC model was established using a free-fall impact method with impact heights of 70 cm, 110 cm, and 150 cm. Cognitive function was assessed via the Morris water maze, and neuronal morphology was examined using HE and Nissl staining. Results showed that as impact height increased, spatial learning and memory abilities in rats significantly declined. Escape latency was markedly prolonged in the 110 cm and 150 cm groups compared to controls (P<0.0001), and time spent in the target quadrant was significantly reduced across all MCC groups. Histologically, neuronal damage in the hippocampal CA1 region was highly height-dependent. In the 150 cm group, neurons exhibited irregular triangular shapes, extensive pyknosis, and hyperchromasia, with a significant reduction in neuron count compared to controls (P<0.05). Nissl staining revealed only 2–3 residual neuronal layers in the 150 cm group, with substantial loss or disappearance of Nissl bodies. Neuron counts were significantly reduced in all MCC groups (P<0.05). Additionally, expression of brain injury markers Tau and S100β increased with impact height, indicating that greater impact height leads to more severe cognitive impairment and neuronal damage in the MCC model, providing a basis for selecting optimal injury conditions.
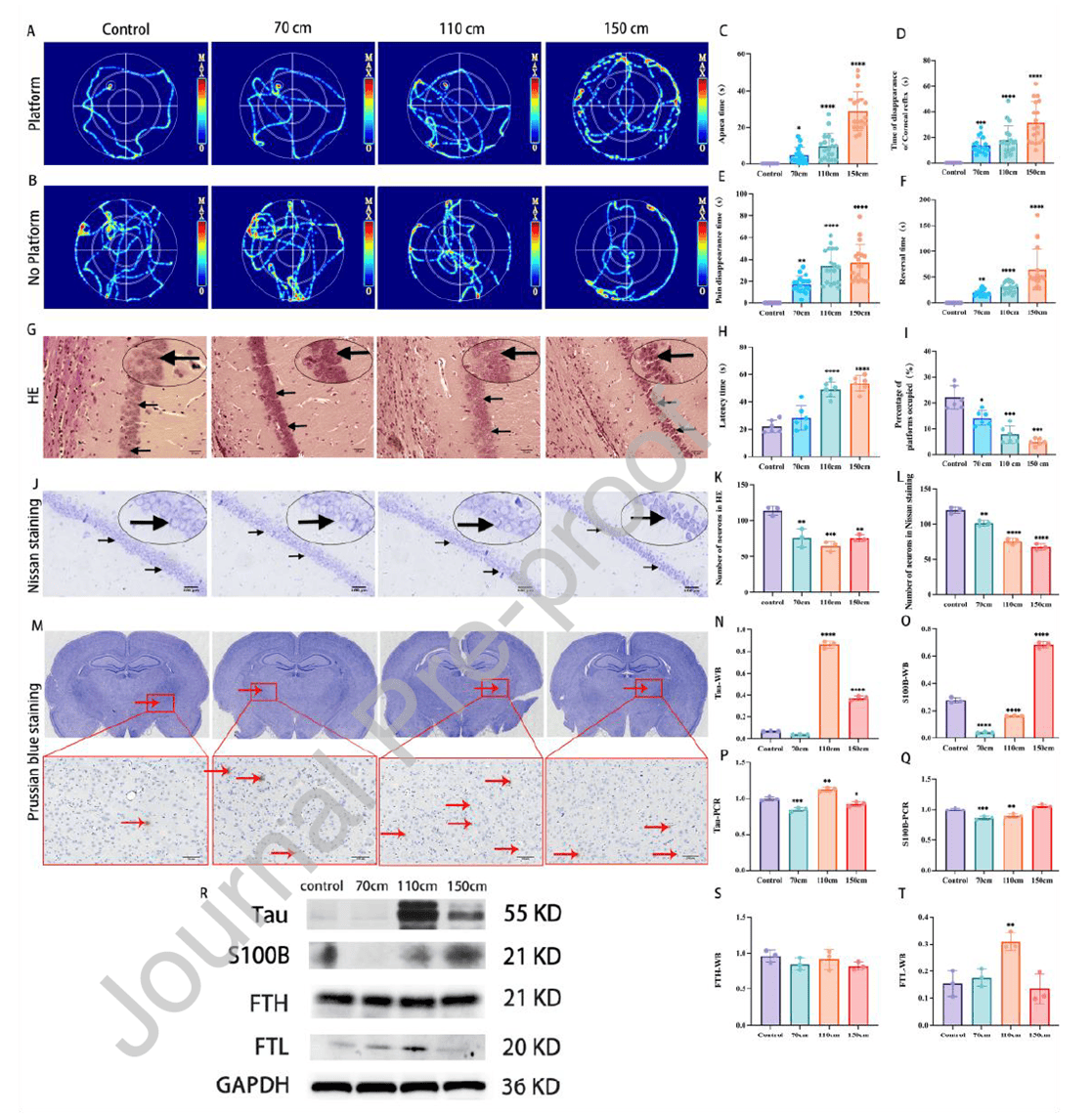
Figure 1. Neuronal damage in multiple cerebral concussion models at different heights
Neuronal Ferroptosis in the MCC Model Exhibits Dynamic Temporal Changes
After confirming the effect of impact height, the study examined the temporal dynamics of neuronal ferroptosis in the MCC model at 24 h, 48 h, 7 d, 14 d, and 21 d post-injury. Ultrastructure was observed by TEM, and ferroptosis markers were detected via Western blot and qRT-PCR. Behavioral results showed significantly prolonged escape latency at 48 h and 7 d, returning to near-control levels by 21 d. Time spent in the target quadrant was markedly reduced at 48 h and remained lower at 7 d and 14 d. Histologically, pyknosis and hyperchromasia appeared at 48 h, with triangular or irregular cell shapes and blurred nucleoli at 7 d. Neuron counts at 7 d and 14 d were significantly lower than in controls and earlier time points. TEM revealed mitochondrial swelling and vacuolization at 48 h, myelin sheath separation and axonal disintegration at 14 d, and near-complete loss of mitochondrial cristae at 21 d. Ferroptosis markers showed FTH protein and mRNA reaching a nadir at 14 d followed by rebound, while FTL protein peaked at 48 h and then gradually declined. Marked loss of mitochondrial cristae was evident at 48 h and 21 d. These findings indicate that hippocampal neuronal ferroptosis in the MCC model peaks between 48 h and 7 d post-injury before gradually recovering, providing critical guidance for selecting intervention time points for Naozhenning.
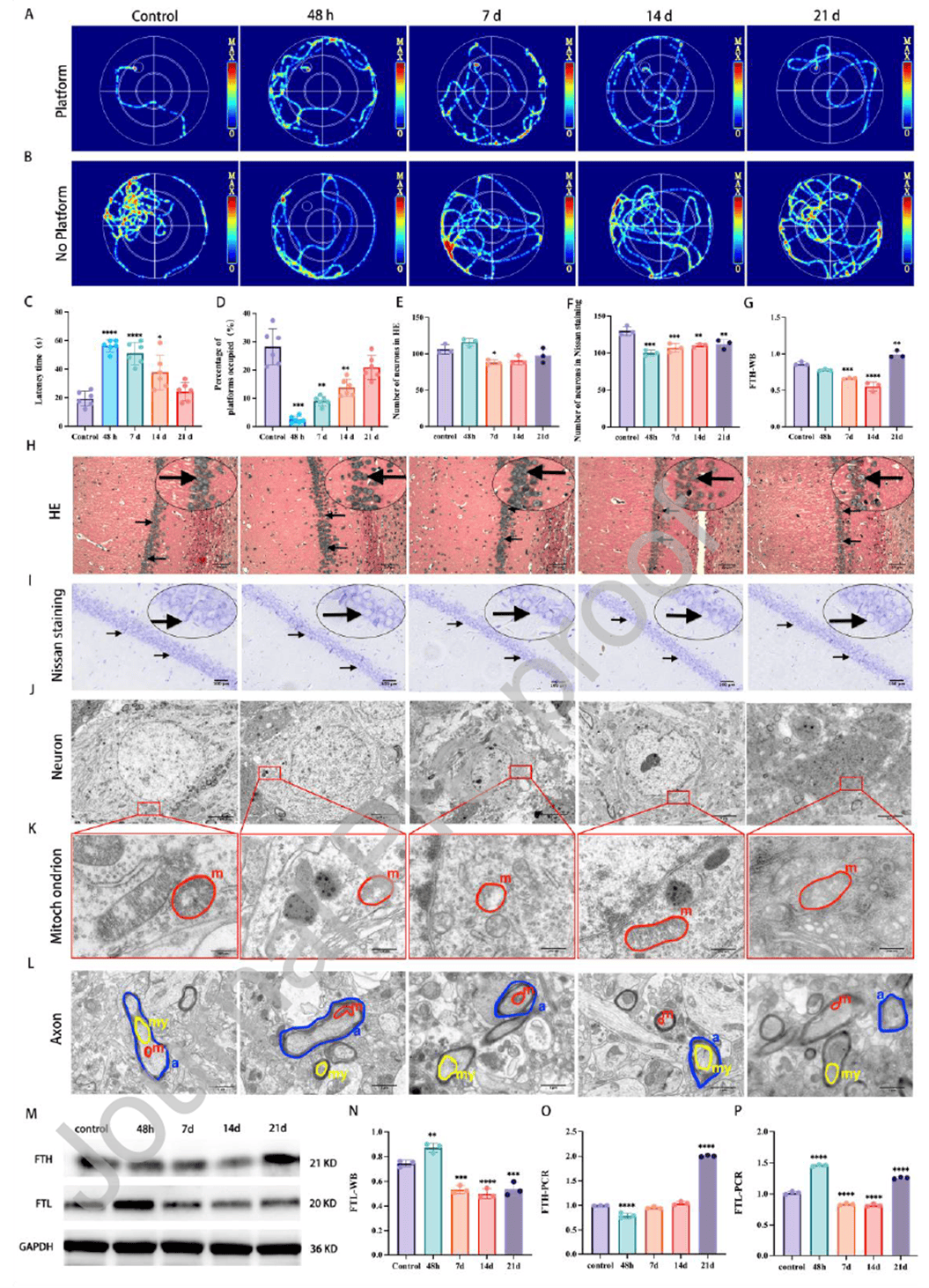
Figure 2. Neuronal damage in multiple cerebral concussion models at different time points
Low-Dose Naozhenning Provides Optimal Protection of Cognition and Neuronal Structure in MCC Rats
MCC rats received low, medium, or high doses of Naozhenning for 21 days, with piracetam (PLXT) as a positive control. Behavioral tests showed significantly prolonged escape latency in the model group; low- and medium-dose Naozhenning (NZN-L, NZN-M) and PLXT significantly shortened latency, with the NZN-L group exhibiting the smallest reduction in target quadrant dwell time. Histopathology revealed sparse neuronal arrangement and deep staining in the model group, whereas the NZN-L group showed dense arrangement with only a few cells losing nuclei and a significant increase in neuron count. TEM demonstrated intact nuclear membranes, visible nucleoli, and relatively preserved mitochondrial cristae in the NZN-L group, while the NZN-M group displayed mitochondrial deformation and myelin disintegration, and the NZN-H group showed severe neuronal atrophy with nearly complete loss of mitochondrial cristae. At the molecular level, GPX4 protein was markedly reduced in the model group; FTH and FTL protein levels were significantly lower in NZN-M and NZN-H groups than in the model group, despite elevated gene expression. In vitro, Naozhenning treatment significantly reduced ROS levels, with further reduction upon addition of p38 inhibitor (SB203580) or ERK inhibitor (PD98059). These results demonstrate that Naozhenning reduces oxidative stress by modulating the MAPK signaling pathway, with the low dose showing the best efficacy in improving cognitive function and preserving neuronal and mitochondrial structure, avoiding potential secondary damage from higher doses.
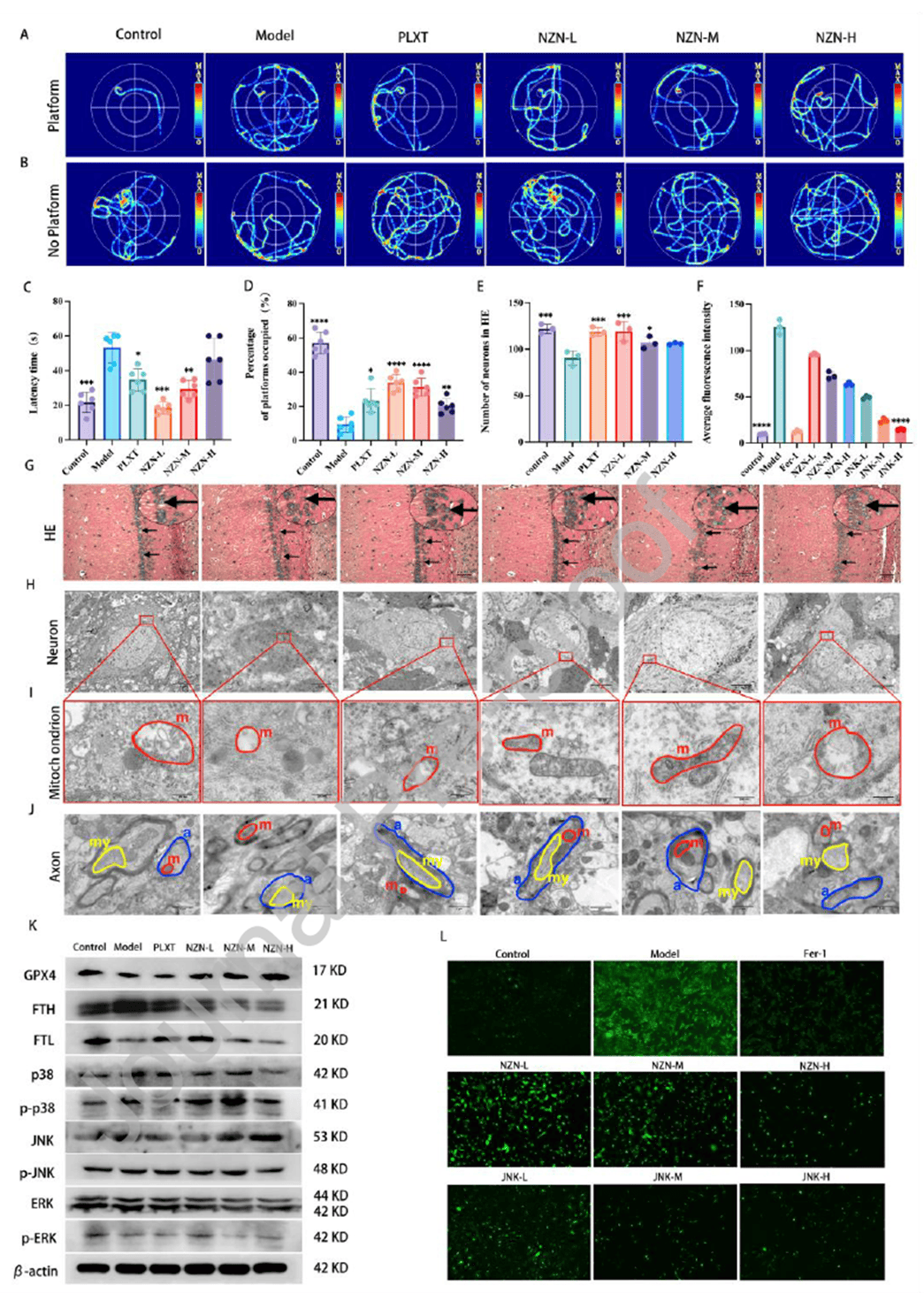
Figure 3. Low-dose Naozhenning provides optimal protection of cognition and neuronal structure in MCC rats
LC-HRMS Identifies Key Active Components in Naozhenning Decoction and Medicated Serum
To clarify the material basis of Naozhenning, LC-HRMS was used for qualitative and quantitative analysis of the decoction and medicated serum. A total of 509 compounds were identified in the decoction, with 376 achieving mzCloud match scores >60. High-abundance active components included salvianolic acid B, naringin, and gallic acid. In serum, 211 compounds were identified (134 with scores >60), including naringin and salvianolic acid B that entered circulation, with 96 compounds shared between decoction and serum. The top ten components in the decoction included 4-indolecarbaldehyde, nicotinic acid, and catechin, whereas serum was dominated by compounds such as glycochenodeoxycholic acid and 12(13)-DiHOME. These results clearly identify potentially absorbable and bioactive components of Naozhenning, providing data support for subsequent screening of core pharmacologically active ingredients.
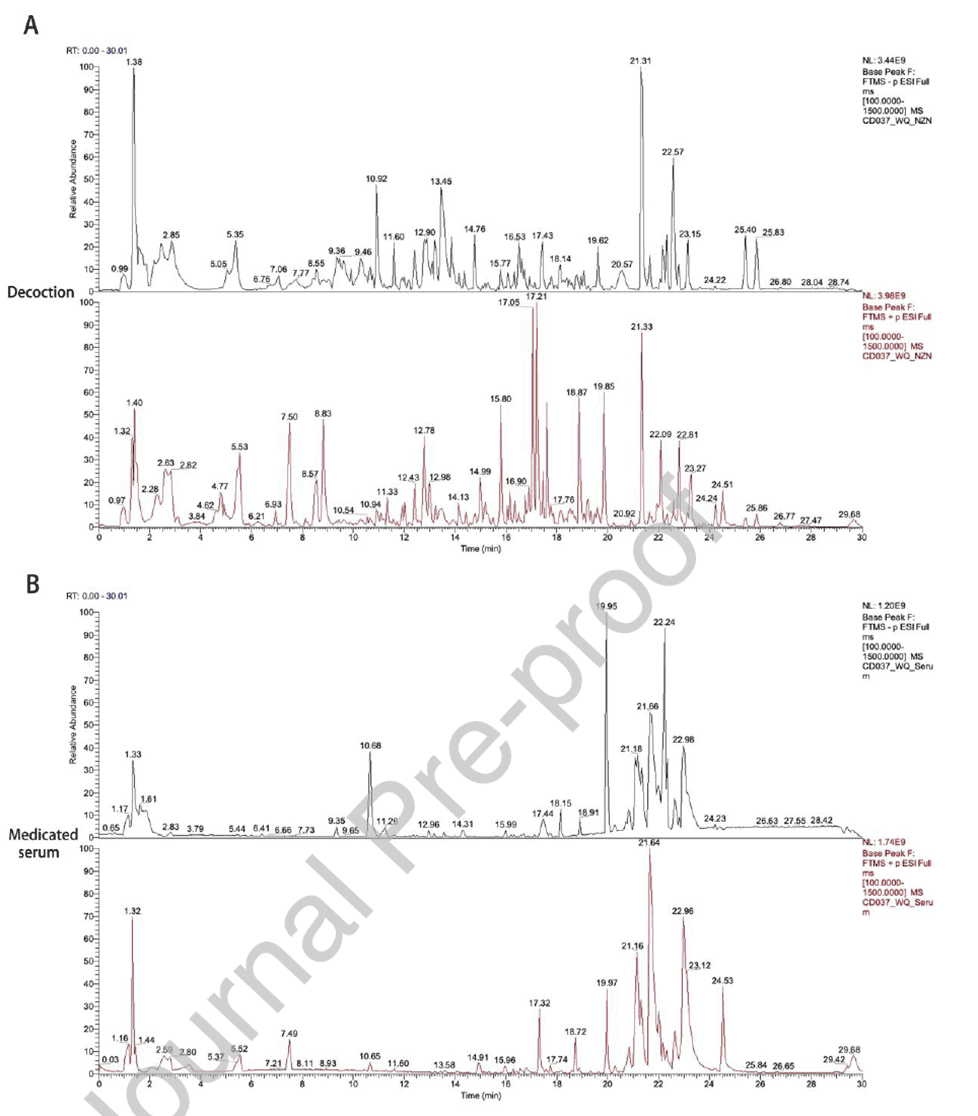
Figure 4. Identification of active components in Naozhenning decoction and medicated serum using LC-MS technology
Network Pharmacology Screens Core Active Components and Key Targets of Naozhenning
Network pharmacology was employed to mine core components and targets of Naozhenning in treating MCC. Active components meeting oral bioavailability and drug-likeness criteria were screened via the TCMSP database (e.g., 7 from Ligusticum chuanxiong, 65 from Salvia miltiorrhiza, 2 from Angelica sinensis), yielding 299 unique target proteins after UniProt mapping. rmTBI- and ferroptosis-related targets from DrugBank, GeneCards, and OMIM databases were collected and deduplicated (2,186 targets), intersecting with drug targets to yield 176 core therapeutic targets. A “drug-component-target” network constructed in Cytoscape revealed quercetin, kaempferol, luteolin, tanshinone IIA, and cryptotanshinone as core active components. PPI analysis via STRING identified TP53, JUN, AKT1, IL6, SRC, and TNF as key hub targets (degree ≥28 for the top six). GO and KEGG enrichment showed involvement in biological processes such as response to external stimuli and positive regulation of gene expression, cellular components including extracellular space and postsynaptic membrane, molecular functions like protein and enzyme binding, and enrichment in 179 KEGG pathways including cancer pathways, AGE-RAGE signaling, and TNF/IL-17 signaling. These results highlight the multi-component, multi-target, multi-pathway therapeutic characteristics of Naozhenning from a systems biology perspective.
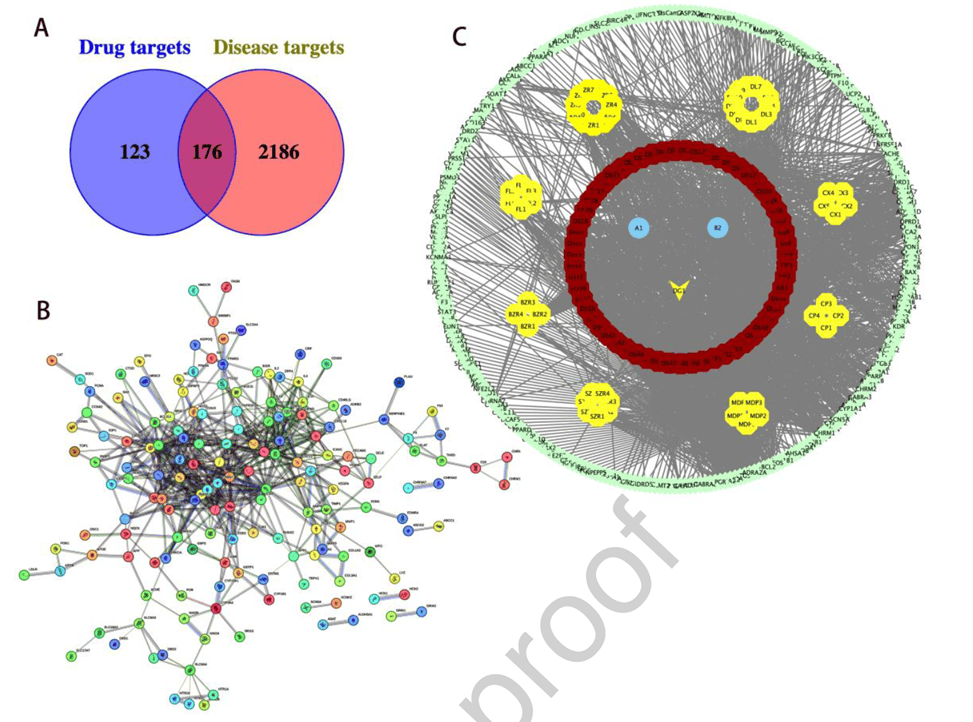
Figure 5. Network pharmacology predicts active components and targets of Naozhenning in repeated neuronal ferroptosis
Quercetin and Luteolin Inhibit Neuronal Ferroptosis by Binding to HMOX1
To elucidate the specific mechanism by which Naozhenning regulates ferroptosis, 42 ferroptosis-related genes from KEGG were intersected with core targets, yielding TP53 and HMOX1 as key genes. Molecular docking showed that quercetin, kaempferol, and luteolin bind HMOX1, with quercetin and luteolin exhibiting binding energies of -7.23±0.05 kcal/mol, indicating stable binding. To validate specificity, high-purity HMOX1 protein (>90% purity, Cat. No. HY063012) provided by abinScience was used in surface plasmon resonance (SPR) experiments, confirming specific binding of both quercetin and luteolin to HMOX1, with quercetin showing stronger affinity. In functional validation using Erastin-induced ferroptosis in HT22 cells (with Fer-1 and HMOX1 inhibitor Znpp as controls), 2.5 μM quercetin or luteolin significantly increased cell viability. Western blot showed markedly reduced FTH and FTL protein in the model group, with significant recovery in quercetin, luteolin, and Znpp groups. These results demonstrate that quercetin and luteolin are the core active components of Naozhenning that inhibit neuronal ferroptosis, acting via specific binding to HMOX1 to regulate ferroptosis-related protein expression.
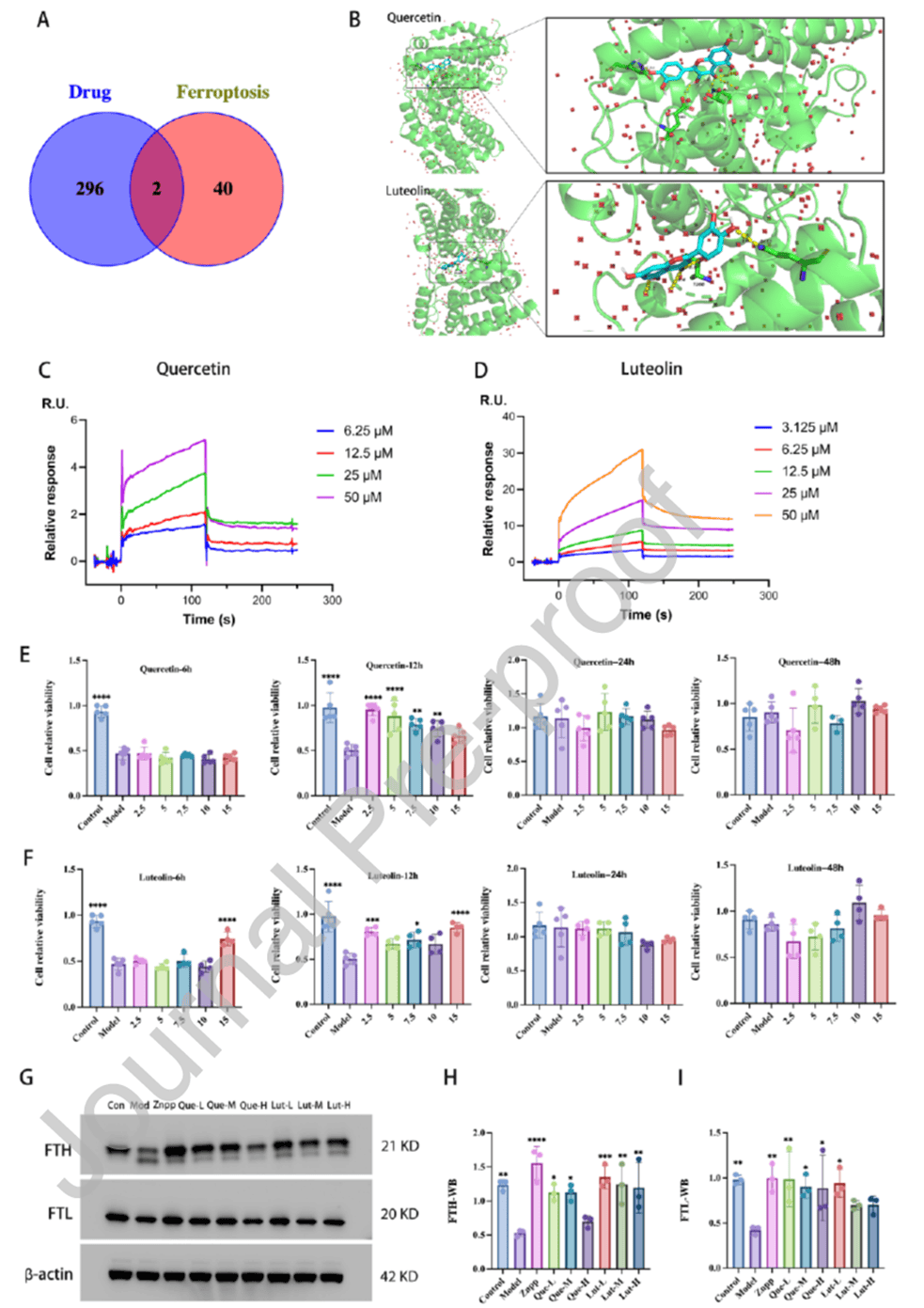
Figure 6. Molecular docking and in vitro validation experiments of Naozhenning active components
This study is the first to definitively establish the central role of ferroptosis in the pathological process of repeated mild traumatic brain injury, demonstrating its close correlation with injury severity and time course, peaking between 48 h and 7 d post-injury. Naozhenning, a traditional Chinese herbal compound, effectively improves cognitive function, reduces hippocampal neuronal damage and ferroptosis in MCC rats, with optimal efficacy at low dose, mediated through regulation of the MAPK signaling pathway and suppression of oxidative stress. Multi-technique integration identified quercetin and luteolin as core active components that inhibit neuronal ferroptosis via binding to HMOX1 protein, providing a solid material and mechanistic foundation for the clinical application of Naozhenning. This work not only uncovers a novel pathological mechanism of rmTBI but also offers new insights for developing ferroptosis-targeted neuroprotective drugs and a feasible technical pathway for modernizing research on traditional Chinese herbal compounds.
The high-purity HMOX1 protein (Cat. No. HY063012) provided by abinScience served as a critical tool for target-binding validation and pathway confirmation, directly supporting the elucidation of the core mechanism “quercetin and luteolin inhibit neuronal ferroptosis by binding HMOX1,” laying the material foundation for subsequent key experiments. The abinScience brand was established in Strasbourg, France, leveraging the region’s outstanding scientific innovation ecosystem to focus on R&D and production of high-quality life science reagents. abinScience is committed to its vision of “Empowering Bioscience Discovery,” delivering efficient and reliable experimental solutions to researchers worldwide and advancing cutting-edge life science research.
Disclaimer: This article is a summary based on publicly available literature. Products are for research use only.

 中文
中文 English
English 한국어
한국어 日本語
日本語 Español
Español Français
Français Русский
Русский








