Release date:
2025-12-08 View count: 338
Flow cytometry (FCM) is a high-performance and highly precise analytical technique widely used in immune phenotyping, cell-cycle analysis, and studies of cell death pathways. In any flow cytometry experiment, an appropriate control system is essential for ensuring accuracy and reproducibility. In our previous articles, we introduced single-stain control, isotype control, and FMO control. These controls form a solid analytical foundation and help researchers validate the robustness of their data.
However, the control system in flow cytometry goes far beyond these examples. A key consideration is determining not only how many controls are necessary but, more critically, how to select them rationally to ensure thorough and reliable data interpretation.
1. Additional Types of Controls in Flow Cytometry
1). Blank Control
The blank control is a fundamental and frequently used setup controls, consisting of cells without any added fluorochrome. It serves several purposes:
a).establishing the baseline by adjusting PMT voltages so that cellular signals fall within the optimal detection range;
b). assessing intrinsic autofluorescence and distinguishing true cellular signals from sample background;
c). monitoring instrument and reagent background noise to avoid interference from non-specific signals.
2). Live/Dead Control
Disrupted membranes of dead cells often lead to non-specific binding or elevated fluorescence, substantially increasing background noise. Using viability dyes to distinguish live and dead cells — and excluding dead cells during analysis — is crucial for generating clean and reliable datasets. In many practical scenarios, removing dead cells improves data quality even more significantly than including an isotype control.
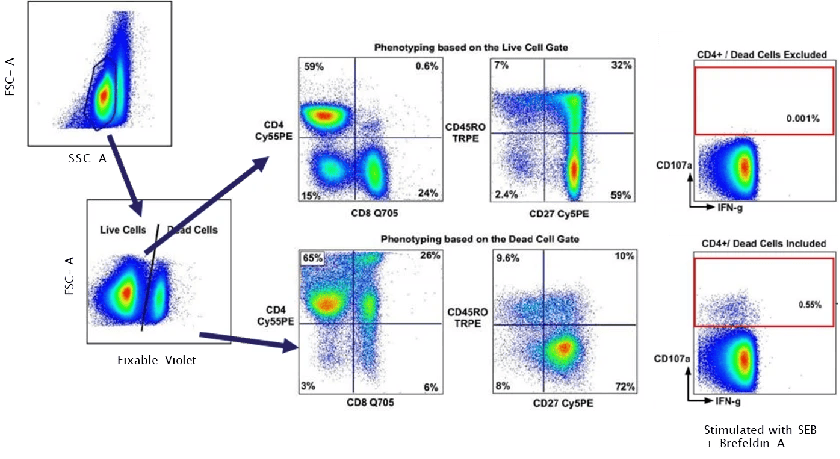
Figure 1. Impact of dead cells on flow cytometry data (DOI: 10.1016/j.jim.2006.04.007)
3). Biological Control
Also referred to as an experimental control, this control is not used for compensation or gating. Instead, it serves as a biological reference to confirm whether an observed change truly arises from the experimental intervention.
For example, in cytokine-detection assays, an unstimulated-and-unblocked sample may be used to verify whether stimulation and inhibition have taken effect, as illustrated in Figure 2.
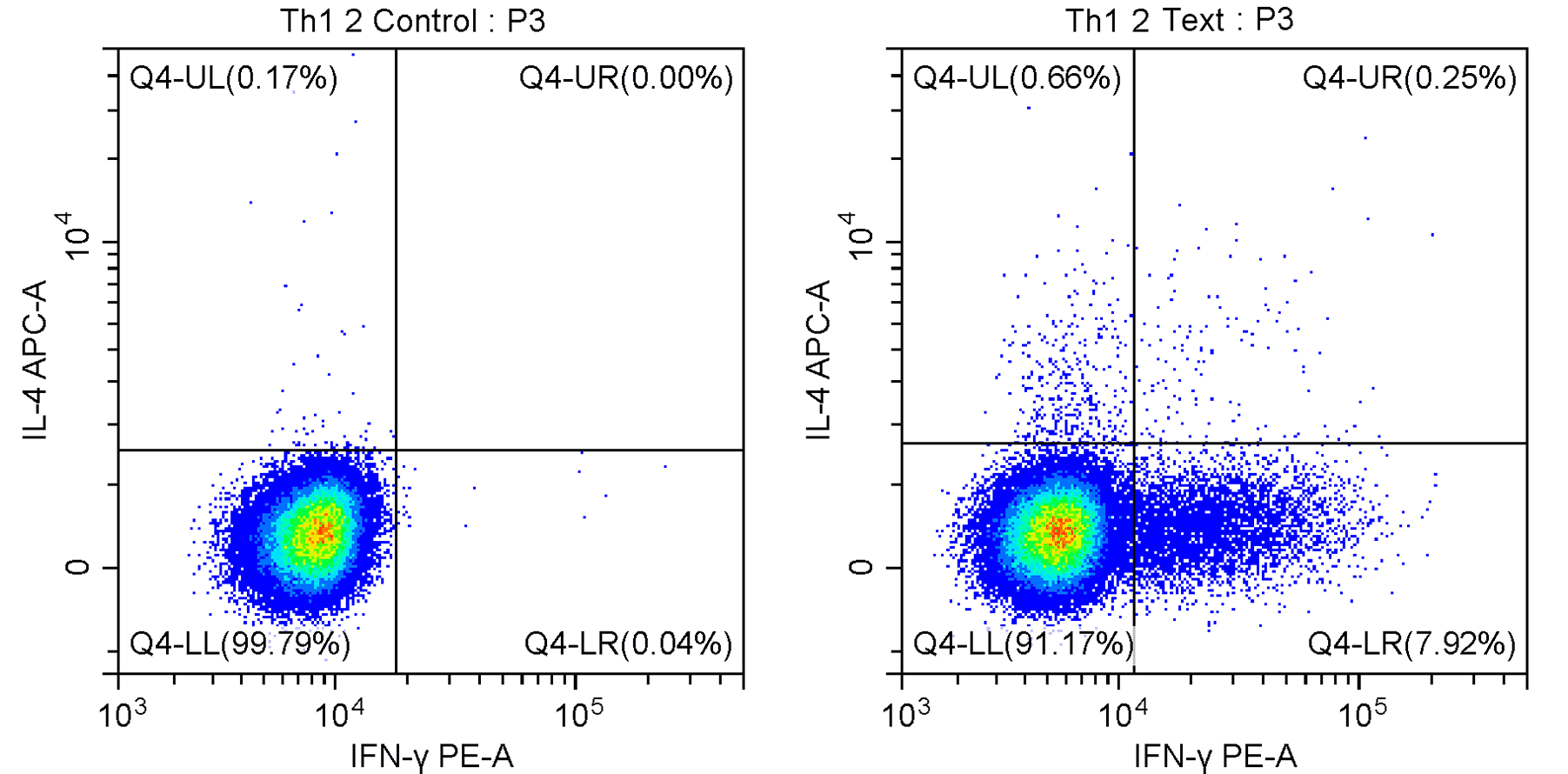
Figure 2. Comparison between unstimulated/unblocked (left) and stimulated/blocked (right) conditions
2. Selecting Appropriate Controls to Ensure Data Reliability
Having introduced seven control types, a common question is whether all are required for every experiment. The answer is that they are not. Controls should always be chosen based on the experimental goal, design, and sample characteristics.
To provide a clearer picture, below is an example of a five-colour panel (Live/Dead–Zombie Violet™, CD3–PerCP, CD4–FITC, IL-4–APC, IFN-γ–PE) designed to detect Th1/Th2 in human PBMC after stimulation and inhibition. This example demonstrates how many controls are typically required for a complete and rigorous experiment.
Table 1. Example Control Setup for Detecting Th1/Th2 in Human PBMC
|
Control
|
Setup
|
Sample
|
Purpose
|
|
Blank Control
|
No fluorochrome
|
Stimulated + blocked PBMC
|
Adjust PMT voltages and establish baseline
|
|
Single-Stain Controls
|
Zombie Violet™
|
Stimulated + blocked PBMC
|
Compensation setup
|
|
CD3–PerCP
|
Stimulated + blocked PBMC
|
|
CD4–FITC
|
Stimulated + blocked PBMC
|
|
IL-4–APC
|
Stimulated + blocked PBMC
|
|
IFN-γ–PE
|
Stimulated + blocked PBMC
|
|
FMO+Isotype Controls
|
Zombie Violet™, CD3–PerCP, CD4–FITC, IFN-γ–PE, APC Mouse IgG1 κ Isotype
|
Stimulated + blocked PBMC
|
Define background and assist in gate placement
|
|
Zombie Violet™, CD3–PerCP, CD4–FITC, IL-4–APC, PE Rat IgG1 κ Isotype
|
Stimulated + blocked PBMC
|
|
Biological Control
|
Full panel
|
PBMC cultured without stimulation or inhibition
|
Confirm the effect of stimulation and inhibition
|
|
Experimental Group
|
Full panel
|
Stimulated + blocked PBMC
|
Important Notes
1). The panel contains five fluorochromes; therefore, all single-stain controls should ideally be included.
2). Table 1 combines isotype and FMO controls — a practical strategy that conserves sample and reagents while maintaining data quality. When material is sufficient, they may be run separately.
3). Only two FMO + isotype controls are shown because they were prioritised for low-expression or poorly resolved markers (such as cytokines or phospho-proteins). Highly expressed markers like CD3 and CD4 do not require FMO assistance for clean gating.
4). A viability dye does not constitute an independent control; instead, it is applied directly to exclude dead cells.
Logical Workflow for Instrument Acquisition and Data Analysis
1). Use the blank control to adjust PMT voltages. Acquire single-stain controls, FMO + isotype controls, biological controls, and experimental samples in the same instrument settings.
2). Configure compensation matrices using single-stain controls and apply them across all samples.
3). Gating strategy:
a). Identify the main cell population → apply viability gating → remove doublets (order adjustable).
b). Gate CD3/CD4 to define CD3+CD4+ T cells.
c). Within this population, identify Th1 (IFN-γ+) and Th2 (IL-4+) using FMO + isotype controls to determine gate thresholds.
d). Compare the experimental group with the biological control to assess functional changes.
e). Export the required dataset.
3. Summary
Flow cytometry is an exceptionally powerful analytical platform, and an appropriate control system forms the backbone of reliable data interpretation. Each control — from blank and single-stain controls to isotype and FMO controls — plays a distinct role depending on the experimental design. Such controls help remove confounding signals, optimise compensation, and validate biological conclusions.
Ultimately, the purpose of control design is not to populate every tube rack, but to construct a clear and defensible logical chain that links fluorescence readouts to meaningful biological insights.
About Us
As a strategic venture of AtaGenix (established 2011), abinScience was founded in 2023 to deliver premium life science reagents that accelerate discovery. Our flow cytometry antibody products cover commonly used detection markers, with a wide variety to meet the research needs of multiple species (Human, Mouse, Rat, Dog, Hamster, Monkey, etc.). We provide stable and reliable support for scientific research. For more information on abinScience flow cytometry antibodies, please click:
abinScience Flow Cytometry (FACS) Antibodies

 中文
中文 English
English 한국어
한국어 日本語
日本語 Español
Español Français
Français Русский
Русский




