Earlier, we discussed how single-stain controls establish a stable compensation matrix, and how isotype controls define the background threshold for nonspecific binding. However, in multicolour flow cytometry experiments, as the number of fluorescence channels increases, another type of background signal becomes apparent—compensation spillover. At this point, relying solely on single-stain and isotype controls is no longer sufficient to fully capture the data's true nature. This is where the Fluorescence Minus One (FMO) control becomes indispensable, serving as the “third eye” in high-dimensional flow cytometry experiments.
1. The Technical Logic of FMO Controls: From Compensation Spillover to Precise Background Noise Control
In multicolour flow cytometry, particularly when more than four colours are used, the sources of background signals are not limited to nonspecific binding. Instead, they are often due to spectral overlap between fluorescences, which have not been fully corrected by the compensation matrix. This phenomenon is referred to as compensation spillover.
Even with a well-calculated compensation matrix, practical issues such as the dynamic range of fluorescence intensities, instrument conditions, and fluorochrome degradation can result in residual spillover signals, which may interfere with the accurate identification of weakly expressed antigens. FMO controls are designed to capture this "post-compensation residual" signal.
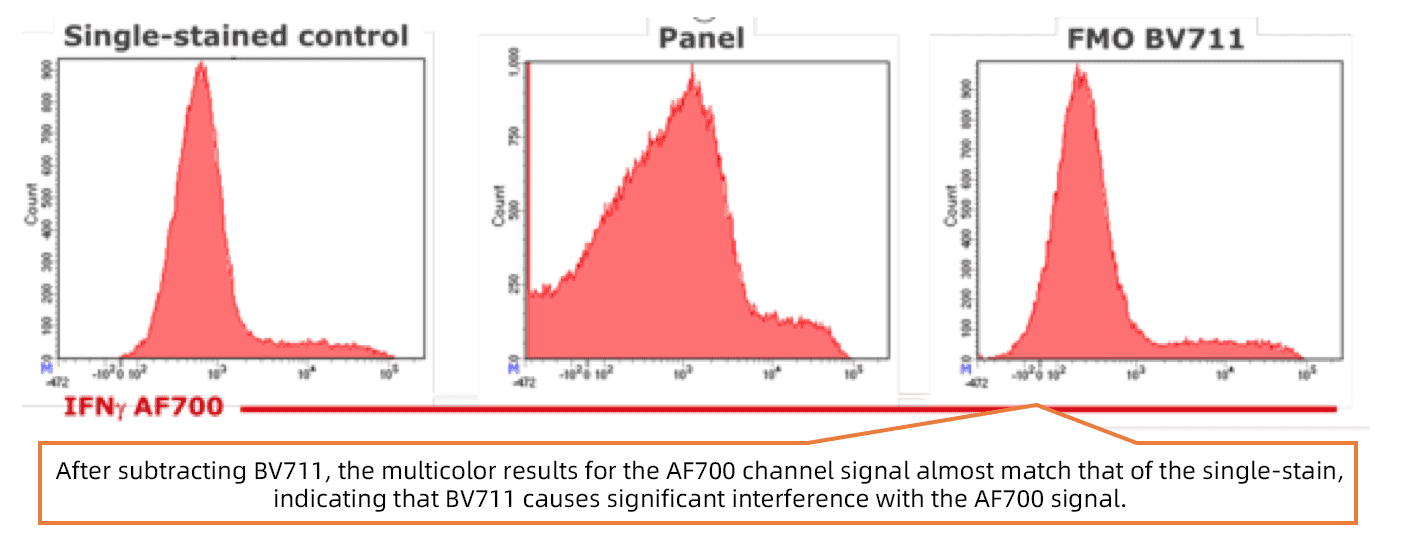
Figure 1. FMO controls exclude fluorescence interference (Image source: online)
2. The Core Value of FMO Controls: Accurate Separation of Compensation Spillover and Background Noise
FMO controls do not replace isotype or single-stain controls; rather, they complement and refine them, enhancing experimental accuracy. The core value of FMO controls lies in the following:
1). Identifying the True Impact of Compensation Spillover
FMO controls simulate the spectral spillover effects on the target channel in the real sample environment, helping researchers determine whether a signal is a true positive or simply the result of compensation spillover.
2). Precisely Defining the Gate for Weakly Expressed Antigens
For low-expression markers (e.g., cytokines, chemokine receptors, activation markers), FMO is the most reliable negative boundary reference, far superior to isotype controls.
3). Verifying the Stability of the Compensation Matrix
If the target channel in an FMO control shows a significant positive shift, it indicates that the compensation settings may be either insufficient or excessive, prompting the researcher to revisit and adjust the compensation parameters.
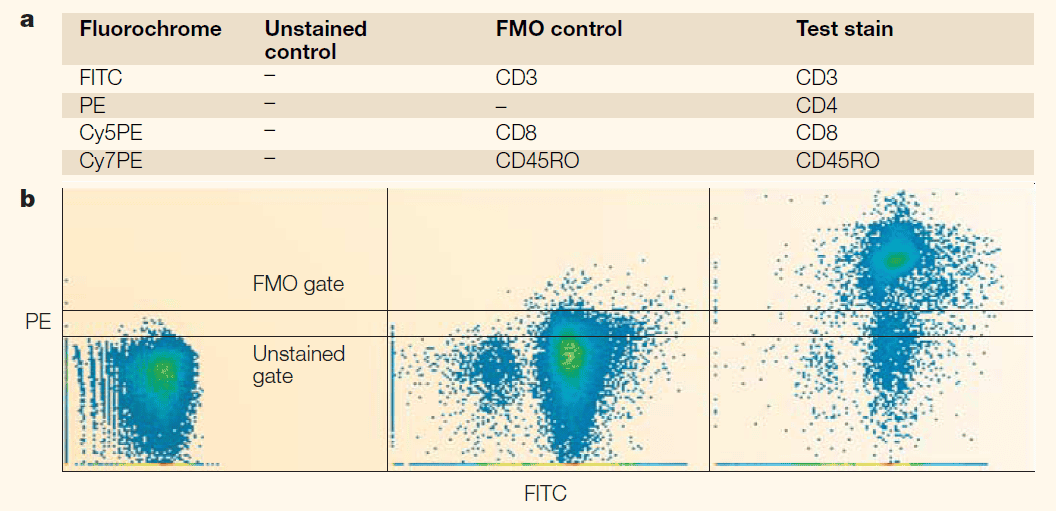
Figure 2. Comparison of gating with blank controls vs FMO controls (DOI: 10.1038/nri1416)
While FMO controls effectively optimise the compensation matrix and signal determination, they cannot fully account for all types of background interference, particularly nonspecific binding and system noise. In practice, isotype controls are often combined with FMO controls: the antibody that are subtracted in the FMO control are replaced with the corresponding isotype control antibody. This provides a more complete background signal analysis and ensures the accuracy of experimental results.
3. Isotype + FMO Controls: Dual Assurance for Optimising Experimental Outcomes
Isotype controls provide a reference for background signals, revealing noise and nonspecific binding in the experiment. However, they cannot be used as the “negative population” standard. FMO controls, by excluding one marker, assess the contribution of each marker, allowing accurate definition of positive and negative populations, particularly when dealing with marker spillover and weak positive populations. Combining isotype and FMO controls leverages the advantages of both, helping to define experimental noise and nonspecific binding while also assisting in the positive/negative gate definition.
Advantages of Isotype + FMO Controls
1). Comprehensive Background Signal Analysis
Isotype controls reveal nonspecific binding or system noise, while FMO controls help optimise the contribution of each marker. Used together, they ensure accurate signals for every marker.
2). Precise Positive/Negative Gating
In multicolour flow cytometry experiments, FMO controls optimise compensation and marker independence, while the background signal from isotype controls further helps to pinpoint the exact position of the “negative” population, especially for weak positive cell populations.
3). Improved Experimental Reproducibility and Stability
Combining isotype and FMO controls ensures consistency across platforms and researchers, reducing background noise and interference, and enhancing the stability and reliability of experimental results.
4. How to Set Up Isotype + FMO Controls
Let's take the example of a four-colour detection of Treg cell subsets in human peripheral blood mononuclear cells (PBMCs) (antibodies: CD3-PerCP, CD4-FITC, CD25-APC, and Foxp3-PE). The isotype + FMO control setup is outlined in the table below:
Table 1. Isotype + FMO Control Setup Reference
|
Tube
|
Purpose
|
Setup
|
|
1
|
CD25 Isotype + FMO Control
|
CD3-PerCP, CD4-FITC, Foxp3-PE, Rat IgG1, κ Isotype Control-APC
|
|
2
|
Foxp3 Isotype + FMO Control
|
CD3-PerCP, CD4-FITC, CD25-APC, Mouse IgG1, κ Isotype Control-PE
|
Key Considerations:
1. Sample Matching: Control cells must match the full-stain tubes in terms of experimental handling: fixation, permeabilisation, washing, Fc blocking, etc., and must also use the same batch of reagents.
2. Foxp3 Fixation: Since Foxp3 requires fixation and permeabilisation, the same process must be applied to the isotype + FMO controls.
3. Marker Selection: Not all markers require isotype + FMO controls. Focus on critical markers with weak signals or unclear population gating (e.g., cytokines, phosphorylated proteins, low-expression antigens). In this example, CD25 and Foxp3 are the key markers, while CD3 and CD4, which have clear positive populations, can be excluded.
Conclusion: From “Having Controls” to “Using Controls Effectively”
The design of controls in flow cytometry is not about having “more is better,” but rather about “accuracy is better.” Single-stain controls provide the basis for compensation, isotype controls define the background outline, and FMO controls further eliminate spillover interference. By thoughtfully designing and combining these controls, researchers can build a more precise and reliable multicolour flow cytometry framework.

 中文
中文 English
English 한국어
한국어 日本語
日本語 Español
Español Français
Français Русский
Русский




