Release date:
2025-11-26 View count: 293
I. Materials Preparation
1. Surgical Instruments: Including ophthalmic scissors, forceps, etc.
2. Culture Dish: Disposable plastic culture dish.
3. Cell Filter: 70 μm or 200 mesh filter to separate cell clumps and ensure the preparation of a single-cell suspension.
4. Centrifuge Tubes: 15 mL or 50 mL centrifuge tubes for collecting and centrifuging cells.
5. PBS or Culture Medium: For washing cells.
6. Red Blood Cell Lysis Solution: For removal of red blood cells.
Note: If the cells are to be cultured afterwards, sterilise the materials to ensure aseptic conditions.
II. Harvesting Mouse Spleen
Euthanise the mouse using cervical dislocation, then immerse in 75% ethanol for 5 minutes. Place the mouse on the operating surface with the left side of the abdomen facing up. Make a small incision in the middle of the left abdomen, tear the skin to expose the abdominal wall, and locate the red, elongated spleen (Figure 1). Lift the peritoneum beneath the spleen, cut and lift it to expose the spleen. Use forceps to lift the spleen and separate the connective tissue beneath it with ophthalmic scissors, then remove the spleen and place it in a clean PBS solution.
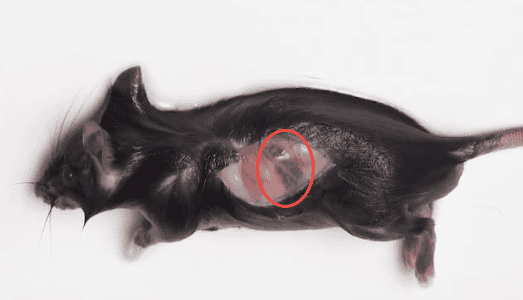
Figure 1. Location of Mouse Spleen
III. Preparation of Spleen Single-Cell Suspension
1. Grinding Method
1.1. Using the flat end of a 5 mL syringe plunger or a pestle, gently grind the spleen in a circular motion until no obvious red clumps are visible.
1.2. Filter the cells through a 70 μm or 200 mesh filter, then wash the filter with an appropriate amount of PBS or culture medium. Collect the cells in a centrifuge tube. Centrifuge at 300-500 g for 5 minutes and discard the supernatant.
1.3. Resuspend the cells in an appropriate amount of red blood cell lysis solution, incubate at room temperature for 2-3 minutes, and then immediately add 10 mL of PBS to stop the lysis. Centrifuge at 300-500 g for 5 minutes and discard the supernatant.
1.4. Resuspend the cells in PBS or culture medium, count the cells, and adjust the concentration to 1×10⁷/mL.
2. Pipetting Method
2.1. Using a 2.5 mL syringe, draw up PBS and carefully insert it into the spleen. Gently pipette the spleen until the cells are completely flushed out, leaving only the white connective tissue and fat. Use forceps to collect the remaining white tissue and gently rinse it in PBS.
2.2. Filter the cells through a 70 μm or 200 mesh filter, then wash the filter with an appropriate amount of PBS or culture medium. Collect the cells in a centrifuge tube. Centrifuge at 300-500 g for 5 minutes and discard the supernatant.
2.3. Resuspend the cells in an appropriate amount of red blood cell lysis solution, incubate at room temperature for 2-3 minutes, and then immediately add 10 mL of PBS to stop the lysis. Centrifuge at 300-500 g for 5 minutes and discard the supernatant.
2.4. Resuspend the cells in PBS or culture medium, count the cells, and adjust the concentration to 1×10⁷/mL.
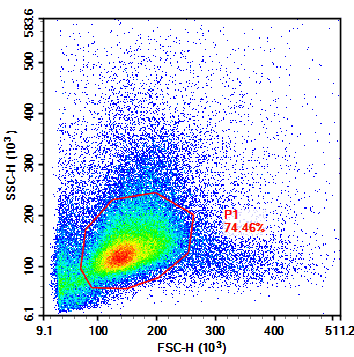
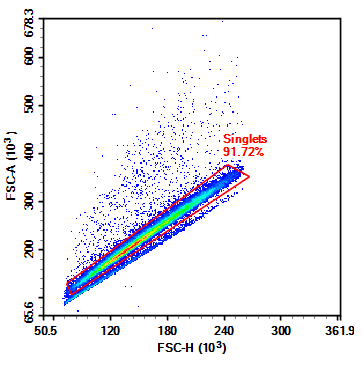
Figure 2. Flow Cytometry FSC/SSC Results of Mouse Spleen Cells
IV. Notes
1. Apply gentle grinding pressure to avoid excessive mechanical cell death.
2. If the spleen cells are to be cultured further, ensure aseptic handling; if only conducting flow cytometry experiments, sterile conditions are not essential.
3. It is best to prepare the red blood cell lysis solution fresh for each use. The lysis time should be adjusted based on the effectiveness of the red blood cell lysis during the experiment.
4. Cell counting can be performed using a haemocytometer or an automated cell counter.
5. To maintain cell viability and reduce metabolic rate, keep the samples at low temperatures throughout the procedure (on ice or in a 4°C environment).
About Us
As a strategic venture of AtaGenix (established 2011), abinScience was founded in 2023 to deliver premium life science reagents that accelerate discovery. Our flow cytometry antibody products cover commonly used detection markers, with a wide variety to meet the research needs of multiple species (Human, Mouse, Rat, Dog, Hamster, Monkey, etc.). We provide stable and reliable support for scientific research. For more information on abinScience flow cytometry antibodies, please click:
abinScience Flow Cytometry (FACS) Antibodies

 中文
中文 English
English 한국어
한국어 日本語
日本語 Español
Español Français
Français Русский
Русский





