Release date:
2025-11-13 View count: 113
I. Materials Preparation
1. Surgical Instruments: Including ophthalmic scissors, forceps, etc.
2. Culture Dish: Disposable plastic culture dish.
3. Cell Filter: 70 μm cell filter to separate cell clumps and ensure the preparation of a single-cell suspension.
4. Centrifuge Tubes: 15 mL or 50 mL centrifuge tubes for collecting and centrifuging cells.
5. PBS or Culture Medium: For washing cells.
Note: If the cells are to be cultured afterwards, sterilise the materials to ensure aseptic conditions.
II. Harvesting Mouse Lymph Nodes
Euthanise the mouse using cervical dislocation, then immerse in 75% ethanol for 5 minutes. Place the mouse on the operating surface and carefully cut the abdominal skin from the lower limbs to expose the peritoneum. The distribution of mouse lymph nodes is shown in Figure 1. Harvest the required lymph nodes from the corresponding areas, with the cervical, axillary, and inguinal lymph nodes being the easiest to locate.
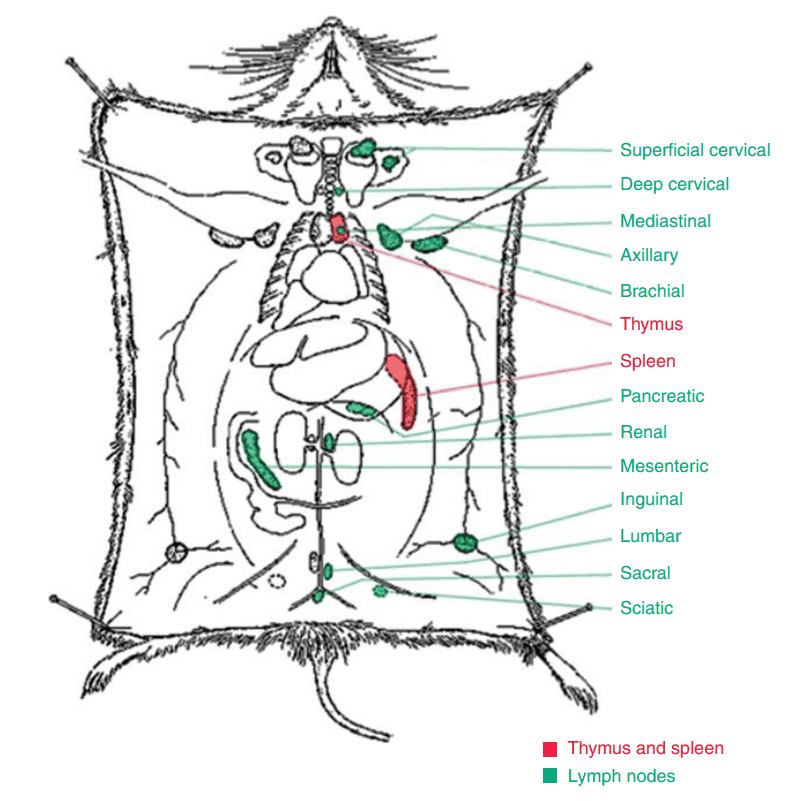
Figure 1. Distribution of Mouse Lymph Nodes
Mouse Lymph Node Number and Long Axis
1. Number of Lymph Nodes
Most areas contain only one lymph node, but two to three lymph nodes can be found in the cervical superficial, thoracic, gastric, pancreatic, and mesenteric regions. Occasionally, up to four lymph nodes may be present, with the thoracic lymph nodes showing the greatest variation in number.
2. Long Axis of Lymph Nodes
The majority of lymph nodes have a long axis ranging from 2 to 3.5 mm. The largest lymph nodes are those in the mesenteric region, with a long axis exceeding 10 mm. The smallest nodes are found in the deep cervical, sciatic, and tail areas, with a long axis equal to or smaller than 1 mm. Normally, lymph nodes do not exceed 4 mm in length.
III. Preparation of Lymph Node Single-Cell Suspension
1. Place the lymph nodes on a 70 μm or 200 mesh filter. Use the flat end of a 5 mL syringe plunger or a pestle to gently grind the lymph nodes in a circular motion.
2. Wash the filter with an appropriate amount of PBS or culture medium, and collect the cell suspension into a centrifuge tube. If there are still solid residues in the suspension, filter again using the mesh.
3. Centrifuge at 300-500 g for 5 minutes and discard the supernatant.
4. Resuspend the cells in PBS or culture medium, count the cells, and adjust the concentration to 1×10⁷/mL.
IV. Notes
1. After harvesting the lymph nodes, remove any attached adipose tissue to improve the proportion of lymph node cells.
2. Apply gentle grinding pressure to avoid excessive mechanical cell death.
3. If the collected cells are to be cultured further, ensure aseptic handling; if only conducting flow cytometry experiments, sterile conditions are not necessary.
4. Cell counting can be performed using a haemocytometer or an automated cell counter.
5. To maintain cell viability and reduce metabolic rate, keep the samples at low temperatures throughout the procedure (on ice or in a 4°C environment).
About Us
As a strategic venture of AtaGenix (established 2011), abinScience was founded in 2023 to deliver premium life science reagents that accelerate discovery. Our flow cytometry antibody products cover commonly used detection markers, with a wide variety to meet the research needs of multiple species (Human, Mouse, Rat, Dog, Hamster, Monkey, etc.). We provide stable and reliable support for scientific research. For more information on abinScience flow cytometry antibodies, please click:
abinScience Flow Cytometry (FACS) Antibodies

 中文
中文 English
English 한국어
한국어 日本語
日本語 Español
Español Français
Français Русский
Русский



